A Pd(II)-Functionalized Covalent Organic Framework for Catalytic Conjugate Additions of Arylboronic Acids to β,β-Disubstituted Enones
- Ames Lab. and Iowa State Univ., Ames, IA (United States)
- Iowa State Univ., Ames, IA (United States)
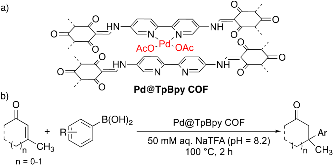
A palladium(II)-functionalized covalent organic framework (Pd@TpBpy COF) built from 1,3,5-triformylphloroglucinol (Tp) and [2,2'-bipyridine]-5,5'-diamine (Bpy) is reported as a recyclable catalyst for conjugate additions in aqueous media. Additions of an array of stereoelectronically diverse arylboronic acid nucleophiles to β,β-disubstituted enones form a variety of ketones containing benzylic all-carbon quaternary centers in up to 92 % isolated yield. Research on the recyclability of Pd@TpBpy COF demonstrate this catalyst remains active through at least 7 cycles and shows superior stability to related MOF catalysts with bipyridine linker units.
- Research Organization:
- Ames Laboratory (AMES), Ames, IA (United States)
- Sponsoring Organization:
- USDOE
- Grant/Contract Number:
- AC02-07CH11358
- OSTI ID:
- 1557818
- Alternate ID(s):
- OSTI ID: 1547943
- Report Number(s):
- IS-J-10004
- Journal Information:
- ChemCatChem, Vol. 11, Issue 17; ISSN 1867-3880
- Publisher:
- ChemPubSoc EuropeCopyright Statement
- Country of Publication:
- United States
- Language:
- English
Cited by: 12 works
Citation information provided by
Web of Science
Web of Science
Similar Records
MOF-253-Pd(OAc)2 : a recyclable MOF for transition-metal catalysis in water
Dioxin-Linked Covalent Organic Framework-Supported Palladium Complex for Rapid Room-Temperature Suzuki–Miyaura Coupling Reaction
Urea-Linked Covalent Organic Frameworks
Journal Article
·
Mon Jun 06 00:00:00 EDT 2016
· RSC Advances
·
OSTI ID:1557818
+1 more
Dioxin-Linked Covalent Organic Framework-Supported Palladium Complex for Rapid Room-Temperature Suzuki–Miyaura Coupling Reaction
Journal Article
·
Thu Aug 17 00:00:00 EDT 2023
· Crystals
·
OSTI ID:1557818
+7 more
Urea-Linked Covalent Organic Frameworks
Journal Article
·
Thu Nov 15 00:00:00 EST 2018
· Journal of the American Chemical Society
·
OSTI ID:1557818
+3 more