Interfacially Induced Cascading Failure in Graphite‐Silicon Composite Anodes
- National Renewable Energy Laboratory 15013 Denver West Parkway Golden CO 80401 USA, Department of Material Science and Engineering Massachusetts Institute of Technology Cambridge MA 02139 USA
- National Renewable Energy Laboratory 15013 Denver West Parkway Golden CO 80401 USA
- National Renewable Energy Laboratory 15013 Denver West Parkway Golden CO 80401 USA, School of Chemical EngineeringYeungnam University Gyeongsan 38541 Republic of Korea
- Department of Material Science and Engineering Massachusetts Institute of Technology Cambridge MA 02139 USA, Electrochemistry BranchSensor and Electron Devices DirectorateU.S. Army Research Laboratory Adelphi MD 20783‐1197 USA
- National Renewable Energy Laboratory 15013 Denver West Parkway Golden CO 80401 USA, Department of Mechanical EngineeringUniversity of Colorado 596 UCB Boulder CO 80309 USA
- ALD NanoSolutions 580 Burbank Street, Unit 100 Broomfield CO 80020 USA
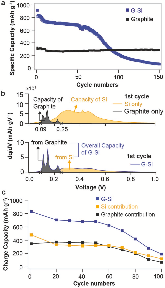
Silicon (Si) has been well recognized as a promising candidate to replace graphite because of its earth abundance and high-capacity storage, but its large volume changes upon lithiation/delithiation and the consequential material fracturing, loss of electrical contact, and over-consumption of the electrolyte prevent its full application. As a countermeasure for rapid capacity decay, a composite electrode of graphite and Si has been adopted by accommodating Si nanoparticles in a graphite matrix. Such an approach, which involves two materials that interact electrochemically with lithium in the electrode, necessitates an analytical methodology to determine the individual electrochemical behavior of each active material. In this work, a methodology comprising differential plots and integral calculus is established to analyze the complicated interplay among the two active batteries and investigate the failure mechanism underlying capacity fade in the blend electrode. To address performance deficiencies identified by this methodology, an aluminum alkoxide (alucone) surface-modification strategy is demonstrated to stabilize the structure and electrochemical performance of the Silicon (Si) has been well recognized as a promising candidate to replace graphite because of its earth abundance and high-capacity storage, but its large volume changes upon lithiation/delithiation and the consequential material fracturing, loss of electrical contact, and over-consumption of the electrolyte prevent its full application. As a countermeasure for rapid capacity decay, a composite
- Research Organization:
- National Renewable Energy Laboratory (NREL), Golden, CO (United States)
- Sponsoring Organization:
- USDOE Office of Energy Efficiency and Renewable Energy (EERE), Vehicle Technologies Office (EE-3V)
- Grant/Contract Number:
- AC36‐08GO28308; AC36-08GO28308
- OSTI ID:
- 1486914
- Alternate ID(s):
- OSTI ID: 1486916; OSTI ID: 1505077
- Report Number(s):
- NREL/JA-5900-71599; 1801007
- Journal Information:
- Advanced Science, Journal Name: Advanced Science Vol. 6 Journal Issue: 3; ISSN 2198-3844
- Publisher:
- WileyCopyright Statement
- Country of Publication:
- Germany
- Language:
- English
Web of Science
Similar Records
Mechanically and Chemically Robust Sandwich-Structured C@Si@C Nanotube Array Li-Ion Battery Anodes
Review—The Lithiation/Delithiation Behavior of Si-Based Electrodes: A Connection between Electrochemistry and Mechanics