Spatial profiling of stapled α–helical peptide ATSP-7041 in mouse whole-body thin tissue sections using droplet-based liquid microjunction surface sampling-HPLC-ESI–MS/MS
Abstract
Here, the application of a fully automated autosampler/HPLC-ESI–MS/MS system for spatially resolved quantitative droplet-based liquid extraction surface sampling/profiling of stapled α–helical peptide ATSP-7041 in mouse whole-body thin tissue sections is reported. 20-μm-thick serial tissue sections of an ATSP-7041 dosed mouse were prepared and the absolute concentration of the targeted peptide was first determined in different organs using 2.3-mm diameter tissue punches, standard bulk tissue extraction protocols, and subsequent HPLC separation and tandem mass spectrometric analysis. The same organs/locations were then analyzed in neighboring tissue sections using the droplet-based surface sampling approach. The observed ATSP-7041 concentration using this method was always significantly lower than that measured by the tissue punch workflow at the same tissue location of a serial section. Calculated extraction efficiencies were 10.7 ± 0.5% (brain), 11.0 ± 3.2% (liver spot 1), 10.7 ± 2.6% (liver spot 2), 15.0 ± 0.6% (lung) and 12.9 ± 0.7% (blood). While these extraction efficiency values were low, they were reproducible within a given organ. This suggests that once the extraction efficiency is established for a given tissue type and drug, the reproducibility of the droplet-based approach could provide a non-labor intensive and high-throughput means to acquire spatially resolved quantitative analysis of multiplemore »
- Authors:
-
- Oak Ridge National Lab. (ORNL), Oak Ridge, TN (United States)
- Merck Research Labs, West Point, PA (United States)
- Publication Date:
- Research Org.:
- Oak Ridge National Lab. (ORNL), Oak Ridge, TN (United States)
- Sponsoring Org.:
- USDOE Office of Science (SC), Basic Energy Sciences (BES)
- OSTI Identifier:
- 1493155
- Grant/Contract Number:
- AC05-00OR22725
- Resource Type:
- Accepted Manuscript
- Journal Name:
- International Journal of Mass Spectrometry
- Additional Journal Information:
- Journal Volume: 437; Journal Issue: C; Journal ID: ISSN 1387-3806
- Publisher:
- Elsevier
- Country of Publication:
- United States
- Language:
- English
- Subject:
- 60 APPLIED LIFE SCIENCES; ATSP-7041; Stapled peptide; Liquid microjunction; Droplet-based liquid extraction; Surface sampling; Autosampler; Spatial distribution
Citation Formats
Kertesz, Vilmos, Vavrek, Marissa, Freddo, Carol, and Van Berkel, Gary J. Spatial profiling of stapled α–helical peptide ATSP-7041 in mouse whole-body thin tissue sections using droplet-based liquid microjunction surface sampling-HPLC-ESI–MS/MS. United States: N. p., 2018.
Web. doi:10.1016/j.ijms.2018.01.005.
Kertesz, Vilmos, Vavrek, Marissa, Freddo, Carol, & Van Berkel, Gary J. Spatial profiling of stapled α–helical peptide ATSP-7041 in mouse whole-body thin tissue sections using droplet-based liquid microjunction surface sampling-HPLC-ESI–MS/MS. United States. https://doi.org/10.1016/j.ijms.2018.01.005
Kertesz, Vilmos, Vavrek, Marissa, Freddo, Carol, and Van Berkel, Gary J. Wed .
"Spatial profiling of stapled α–helical peptide ATSP-7041 in mouse whole-body thin tissue sections using droplet-based liquid microjunction surface sampling-HPLC-ESI–MS/MS". United States. https://doi.org/10.1016/j.ijms.2018.01.005. https://www.osti.gov/servlets/purl/1493155.
@article{osti_1493155,
title = {Spatial profiling of stapled α–helical peptide ATSP-7041 in mouse whole-body thin tissue sections using droplet-based liquid microjunction surface sampling-HPLC-ESI–MS/MS},
author = {Kertesz, Vilmos and Vavrek, Marissa and Freddo, Carol and Van Berkel, Gary J.},
abstractNote = {Here, the application of a fully automated autosampler/HPLC-ESI–MS/MS system for spatially resolved quantitative droplet-based liquid extraction surface sampling/profiling of stapled α–helical peptide ATSP-7041 in mouse whole-body thin tissue sections is reported. 20-μm-thick serial tissue sections of an ATSP-7041 dosed mouse were prepared and the absolute concentration of the targeted peptide was first determined in different organs using 2.3-mm diameter tissue punches, standard bulk tissue extraction protocols, and subsequent HPLC separation and tandem mass spectrometric analysis. The same organs/locations were then analyzed in neighboring tissue sections using the droplet-based surface sampling approach. The observed ATSP-7041 concentration using this method was always significantly lower than that measured by the tissue punch workflow at the same tissue location of a serial section. Calculated extraction efficiencies were 10.7 ± 0.5% (brain), 11.0 ± 3.2% (liver spot 1), 10.7 ± 2.6% (liver spot 2), 15.0 ± 0.6% (lung) and 12.9 ± 0.7% (blood). While these extraction efficiency values were low, they were reproducible within a given organ. This suggests that once the extraction efficiency is established for a given tissue type and drug, the reproducibility of the droplet-based approach could provide a non-labor intensive and high-throughput means to acquire spatially resolved quantitative analysis of multiple samples of the same type.},
doi = {10.1016/j.ijms.2018.01.005},
journal = {International Journal of Mass Spectrometry},
number = C,
volume = 437,
place = {United States},
year = {Wed Jan 10 00:00:00 EST 2018},
month = {Wed Jan 10 00:00:00 EST 2018}
}
Web of Science
Figures / Tables:
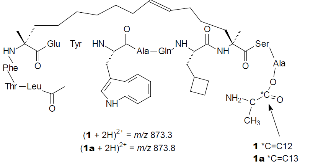 Scheme 1: Structure and mass-to-charge ratio ($m/z$) of major parent ion for ATSP-7041 (1) and internal standard ATSP-7041-C13 (1a).
Scheme 1: Structure and mass-to-charge ratio ($m/z$) of major parent ion for ATSP-7041 (1) and internal standard ATSP-7041-C13 (1a).
Figures / Tables found in this record:

 Search WorldCat to find libraries that may hold this journal
Search WorldCat to find libraries that may hold this journal