Kinesin motor density and dynamics in gliding microtubule motility
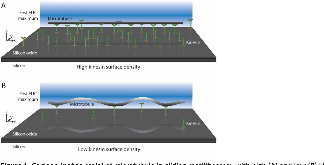
Kinesin motors and their associated filaments, microtubules, are essential to many biological processes. The motor and filament system can be reconstituted in vitro with the surface-adhered motors transporting the filaments along the surface. In this format, the system has been used to study active self-assembly and to power microdevices or perform analyte detection. Yet, fundamental properties of the system, such as the spacing of the kinesin motors bound to the microtubule and the dynamics of binding, remain poorly understood. We show that Fluorescence Interference Contrast (FLIC) microscopy can illuminate the exact height of the microtubule, which for a sufficiently low surface density of kinesin, reveals the locations of the bound motors. We investigate the spacing of the kinesin motors on the microtubules at various kinesin surface densities and compare the results with theory. FLIC reveals that the system is highly dynamic, with kinesin binding and unbinding along the length of the microtubule as it is transported along the surface.
- Research Organization:
- Sandia National Lab. (SNL-NM), Albuquerque, NM (United States)
- Sponsoring Organization:
- USDOE Office of Science (SC), Basic Energy Sciences (BES)
- Grant/Contract Number:
- AC04-94AL85000
- OSTI ID:
- 1619514
- Alternate ID(s):
- OSTI ID: 1515204
- Report Number(s):
- SAND-2019-5346J; 7206; PII: 43749
- Journal Information:
- Scientific Reports, Journal Name: Scientific Reports Vol. 9 Journal Issue: 1; ISSN 2045-2322
- Publisher:
- Nature Publishing GroupCopyright Statement
- Country of Publication:
- United Kingdom
- Language:
- English
Web of Science
Similar Records
Multicomponent and Multiphase Lipid Nanotubes Formed by Gliding Microtubule-Kinesin Motility and Phase-Separated Giant Unilamellar Vesicles
Mechanical splitting of microtubules into protofilament bundles by surface-bound kinesin-1