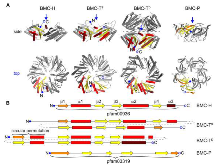
The Plasticity of Molecular Interactions Governs Bacterial Microcompartment Shell Assembly
Bacterial microcompartments (BMCs) are composed of an enzymatic core encapsulated by a selectively permeable protein shell that enhances catalytic efficiency. Many pathogenic bacteria derive a competitive advantage from their BMC based catabolism, implicating BMCs as promising drug targets. BMC shells are of interest for bioengineering due to their diverse and selective permeability properties and because they self-assemble. A complete understanding of shell composition and organization is a prerequisite for biotechnological applications. Here, we report the cryo-EM structure of a BMC shell at 3.0 32 Å resolution, using an image processing strategy that allowed us to determine the previously uncharacterized structural details of the interactions of the BMC-TS and BMC-TD shell subunits with the surrounding BMC-H subunits in the context of the assembled shell. We found unexpected structural plasticity among these interactions, resulting in distinct shell populations assembled from varying numbers of the BMC-TS and BMC-TD shell subunits. Here, our results show that heterologously produced shells are surprisingly compositonally diverse; we discuss the implications of these findings on shell assembly and function.
- Research Organization:
- Michigan State Univ., East Lansing, MI (United States)
- Sponsoring Organization:
- USDOE Office of Science (SC), Basic Energy Sciences (BES); National Institutes of Health, National Institute of Allergy and Infectious Diseases (NIAID)
- Grant/Contract Number:
- FG02-91ER20021; R01 AI114975-05; AC02-05CH11231
- OSTI ID:
- 1617538
- Alternate ID(s):
- OSTI ID: 1671308
- Journal Information:
- Structure, Journal Name: Structure Vol. 27 Journal Issue: 5; ISSN 0969-2126
- Publisher:
- ElsevierCopyright Statement
- Country of Publication:
- United Kingdom
- Language:
- English
Web of Science
Engineering the PduT shell protein to modify the permeability of the 1,2-propanediol microcompartment of Salmonella
|
journal | December 2019 |
Functionalization of Bacterial Microcompartment Shell Proteins With Covalently Attached Heme
|
journal | January 2020 |
Similar Records
A designed bacterial microcompartment shell with tunable composition and precision cargo loading
Glycyl Radical Enzyme-Associated Microcompartments: Redox-Replete Bacterial Organelles