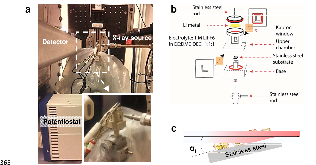
Nanoscale in situ detection of nucleation and growth of Li electrodeposition at various current densities
- Washington Univ., St. Louis, MO (United States). Dept. of Energy, Environmental and Chemical Engineering
- Argonne National Lab. (ANL), Argonne, IL (United States). X-ray Science Division
- Seoul National Univ. (Korea, Republic of). Program in Nano Science and Technology, Graduate School of Convergence Science and Technology
Li metal batteries can store at least ten times more energy than currently existing Li-ion batteries. However, during routine charging and discharging, Li dendrites grow on the Li metal electrode, which can lead to capacity loss by the consumption of Li salt at the surface of the Li dendrites, and be a safety hazard resulting from the potential for short-circuits. Although past efforts have provided useful information about the morphology and surface area of Li dendrite formation at the microscale, a nanoscale understanding of nucleation and growth of Li nanoparticle electrodeposition is still elusive. In this study, using a new electrochemical cell for transmission mode grazing incidence small angle X-ray scattering, we obtained, for the first time, the primary nucleus size of Li nanoparticles, their size evolution and their fractal structures at various current densities and in real-time. The measured average radius of gyration, Rg, at current densities of 0.1, 0.5, and 2.0 mA cm-2 is 5.4 ± 0.4, 4.5 ± 0.3, and 3.5 ± 0.3 nm, respectively. This variation in size with current density is noteworthy when recognizing that the surface area-to-volume ratio of the Li nanoparticles is 3.7 times higher at 2.0 mA cm-2 than at 0.1 mA cm-2. We also compared a hierarchical fractal structure of Li particles from the nanometer to micrometer scale. Our findings illuminate the role of overpotential in the reactive surface area of Li dendrites at the nanoscale, and provide a novel research platform for suppressing Li dendrite formation in Li metal battery systems.
- Research Organization:
- Argonne National Lab. (ANL), Argonne, IL (United States)
- Sponsoring Organization:
- USDOE Office of Science (SC), Basic Energy Sciences (BES); National Science Foundation (NSF)
- Grant/Contract Number:
- AC02-06CH11357
- OSTI ID:
- 1460946
- Journal Information:
- Journal of Materials Chemistry. A, Vol. 6, Issue 11; ISSN 2050-7488
- Publisher:
- Royal Society of ChemistryCopyright Statement
- Country of Publication:
- United States
- Language:
- English
Web of Science
Favorable lithium deposition behaviors on flexible carbon microtube skeleton enable a high-performance lithium metal anode
|
journal | January 2018 |
An acetylene black modified gel polymer electrolyte for high-performance lithium–sulfur batteries
|
journal | January 2019 |
Similar Records
High Current Cycling in a Superconcentrated Ionic Liquid Electrolyte to Promote Uniform Li Morphology and a Uniform LiF-Rich Solid Electrolyte Interphase
Interface Design for High‐Performance All‐Solid‐State Lithium Batteries