Effect of overcharge on Li(Ni0.5Mn0.3Co0.2)O2 cathodes: NMP-soluble binder. II — Chemical changes in the anode
Journal Article
·
· Journal of Power Sources
- Argonne National Lab. (ANL), Argonne, IL (United States). Chemical Sciences and Engineering Division
- Argonne National Lab. (ANL), Argonne, IL (United States). Analytical Chemistry Lab. and Nuclear Engineering Division
- Oak Ridge National Lab. (ORNL), Oak Ridge, TN (United States). Energy and Transportation Science Division
- Sandia National Lab. (SNL-NM), Albuquerque, NM (United States). Power Sources Technology Group
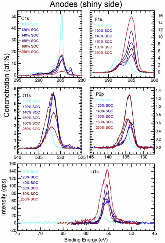
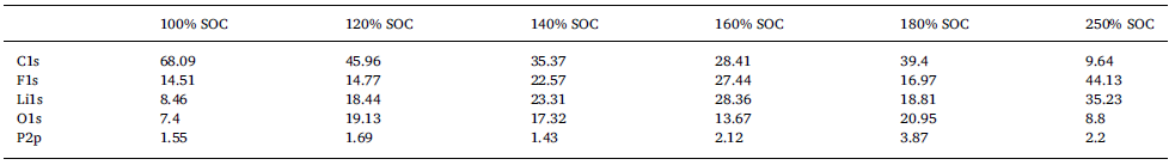
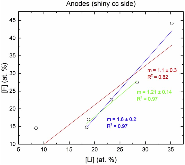
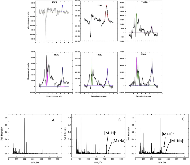
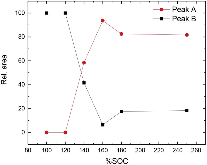
For this study, cells based on nickel manganese cobalt oxide (NMC)/graphite electrodes, which contained polyvinylidene difluoride (PVDF) binders in the electrodes, were systematically charged to 100, 120, 140, 160, 180, and 250% state of charge (SOC). Characterization of the anodes by inductively-coupled-plasma mass spectrometry (ICP-MS), X-ray photoelectron spectroscopy (XPS), and high-performance liquid chromatography coupled with electrospray ionization mass spectrometry (HPLC-ESI-MS) showed several extent-of-overcharge-dependent trends. The concentrations (by wt) of nickel, manganese, and cobalt in the negative electrode increased with SOC, but the metals remained in the same ratio as that of the positive. Electrolyte reaction products, such as LiF:LiPO3, increased with overcharge, as expected. Three organic products were found by HPLC-ESI-MS. From an analysis of the mass spectra, two of these compounds seem to be organophosphates, which were formed by the reaction of polymerized electrolyte decomposition products and PF3 or O=PF3. Their concentration tended to reach a constant ratio. The third was seen at 250% SOC only.
- Research Organization:
- Argonne National Lab. (ANL), Argonne, IL (United States); Argonne National Laboratory (ANL), Argonne, IL (United States); Oak Ridge National Laboratory (ORNL), Oak Ridge, TN (United States); Sandia National Laboratories (SNL-NM), Albuquerque, NM (United States)
- Sponsoring Organization:
- USDOE; USDOE National Nuclear Security Administration (NNSA); USDOE Office of Energy Efficiency and Renewable Energy (EERE), Vehicle Technologies Office (EE-3V)
- Grant/Contract Number:
- AC02-06CH11357; AC05-00OR22725; NA0003525
- OSTI ID:
- 1437886
- Alternate ID(s):
- OSTI ID: 1548669
OSTI ID: 1474299
- Journal Information:
- Journal of Power Sources, Journal Name: Journal of Power Sources Journal Issue: C Vol. 385; ISSN 0378-7753
- Publisher:
- ElsevierCopyright Statement
- Country of Publication:
- United States
- Language:
- English
Overcharge Cycling Effect on the Surface Layers and Crystalline Structure of LiFePO4 Cathodes of Li-Ion Batteries
|
journal | December 2019 |
Chromatographic Techniques in the Research Area of Lithium Ion Batteries: Current State-of-the-Art
|
journal | May 2019 |
Similar Records
Effect of overcharge on Li(Ni0.5Mn0.3Co0.2)O2/Graphite lithium ion cells with poly(vinylidene fluoride) binder. I - Microstructural changes in the anode
Effect of overcharge on Li(Ni0.5Mn0.3Co0.2)O2/graphite lithium ion cells with poly(vinylidene fluoride) binder. III — Chemical changes in the cathode
Journal Article
·
Fri Mar 16 20:00:00 EDT 2018
· Journal of Power Sources
·
OSTI ID:1439931
Effect of overcharge on Li(Ni0.5Mn0.3Co0.2)O2/graphite lithium ion cells with poly(vinylidene fluoride) binder. III — Chemical changes in the cathode
Journal Article
·
Tue Jan 16 19:00:00 EST 2018
· Journal of Power Sources
·
OSTI ID:1437885