CO2/brine/rock interactions in Lower Tuscaloosa formation
Abstract
Saline aquifers are the largest potential continental geologic CO2 sequestration resource. Understanding of potential geochemically induced changes to the porosity and permeability of host CO2 storage and sealing formation rock will improve our ability to predict CO2 plume dynamics, storage capacity, and long–term reservoir behavior. Experiments exploring geochemical interactions of CO2/brine/rock on saline formations under CO2 sequestration conditions were conducted in a static system. Chemical interactions in core samples from the Lower Tuscaloosa formation from Jackson County, Mississippi, with exposure to CO2–saturated brine under sequestration conditions were studied through six months of batch exposure. The experimental conditions to which the core samples of Lower Tuscaloosa sandstone and Selma chalk were exposed to a temperature of 85°C, CO2 pressure of 23.8 MPa (3500 psig), while immersed in a model brine representative of Tuscaloosa Basin. Computed tomography (CT), X–Ray diffraction (XRD), Scanning Electron Microscopy (SEM), brine chemistry, and petrography analyses were performed before and after the exposure. Permeability measurements from the sandstone core sample before and after exposure showed a permeability reduction. No significant change of the permeability measurements was noticed for the core sample obtained from Selma chalk after it was exposed to CO2/brine for six months. Furthermore, these results havemore »
- Authors:
-
- National Energy Technology Lab. (NETL), Pittsburgh, PA (United States)
- Publication Date:
- Research Org.:
- National Energy Technology Laboratory (NETL), Pittsburgh, PA, Morgantown, WV, and Albany, OR (United States)
- Sponsoring Org.:
- USDOE
- OSTI Identifier:
- 1569756
- Report Number(s):
- NETL-PUB-20283
Journal ID: ISSN 2152-3878
- Resource Type:
- Accepted Manuscript
- Journal Name:
- Greenhouse Gases: Science and Technology
- Additional Journal Information:
- Journal Volume: 6; Journal Issue: 6; Journal ID: ISSN 2152-3878
- Publisher:
- Society of Chemical Industry, Wiley
- Country of Publication:
- United States
- Language:
- English
- Subject:
- 58 GEOSCIENCES; CO2 sequestration; Tuscaloosa formation; chemical interaction; permeability; saline aquifer
Citation Formats
Soong, Yee, Howard, Bret H., Dilmore, Robert M., Haljasmaa, Igor, Crandall, Dustin M., Zhang, Liwei, Zhang, Wu, Lin, Ronghong, Irdi, Gino A., Romanov, Vyacheslav N., and Mclendon, Thomas R. CO2/brine/rock interactions in Lower Tuscaloosa formation. United States: N. p., 2016.
Web. doi:10.1002/ghg.1611.
Soong, Yee, Howard, Bret H., Dilmore, Robert M., Haljasmaa, Igor, Crandall, Dustin M., Zhang, Liwei, Zhang, Wu, Lin, Ronghong, Irdi, Gino A., Romanov, Vyacheslav N., & Mclendon, Thomas R. CO2/brine/rock interactions in Lower Tuscaloosa formation. United States. https://doi.org/10.1002/ghg.1611
Soong, Yee, Howard, Bret H., Dilmore, Robert M., Haljasmaa, Igor, Crandall, Dustin M., Zhang, Liwei, Zhang, Wu, Lin, Ronghong, Irdi, Gino A., Romanov, Vyacheslav N., and Mclendon, Thomas R. Wed .
"CO2/brine/rock interactions in Lower Tuscaloosa formation". United States. https://doi.org/10.1002/ghg.1611. https://www.osti.gov/servlets/purl/1569756.
@article{osti_1569756,
title = {CO2/brine/rock interactions in Lower Tuscaloosa formation},
author = {Soong, Yee and Howard, Bret H. and Dilmore, Robert M. and Haljasmaa, Igor and Crandall, Dustin M. and Zhang, Liwei and Zhang, Wu and Lin, Ronghong and Irdi, Gino A. and Romanov, Vyacheslav N. and Mclendon, Thomas R.},
abstractNote = {Saline aquifers are the largest potential continental geologic CO2 sequestration resource. Understanding of potential geochemically induced changes to the porosity and permeability of host CO2 storage and sealing formation rock will improve our ability to predict CO2 plume dynamics, storage capacity, and long–term reservoir behavior. Experiments exploring geochemical interactions of CO2/brine/rock on saline formations under CO2 sequestration conditions were conducted in a static system. Chemical interactions in core samples from the Lower Tuscaloosa formation from Jackson County, Mississippi, with exposure to CO2–saturated brine under sequestration conditions were studied through six months of batch exposure. The experimental conditions to which the core samples of Lower Tuscaloosa sandstone and Selma chalk were exposed to a temperature of 85°C, CO2 pressure of 23.8 MPa (3500 psig), while immersed in a model brine representative of Tuscaloosa Basin. Computed tomography (CT), X–Ray diffraction (XRD), Scanning Electron Microscopy (SEM), brine chemistry, and petrography analyses were performed before and after the exposure. Permeability measurements from the sandstone core sample before and after exposure showed a permeability reduction. No significant change of the permeability measurements was noticed for the core sample obtained from Selma chalk after it was exposed to CO2/brine for six months. Furthermore, these results have implications for performance of the storage interval, and the integrity of the seal in a CO2 storage setting.},
doi = {10.1002/ghg.1611},
journal = {Greenhouse Gases: Science and Technology},
number = 6,
volume = 6,
place = {United States},
year = {Wed Dec 07 00:00:00 EST 2016},
month = {Wed Dec 07 00:00:00 EST 2016}
}
Web of Science
Figures / Tables:
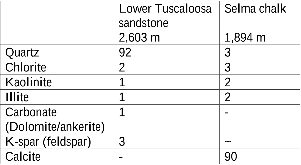 Table 1: XRD analysis of the Lower Tuscaloosa sandstone and the Selma chalk carbonate rock. (Analysis was performed by Weatherford Laboratories.)
Table 1: XRD analysis of the Lower Tuscaloosa sandstone and the Selma chalk carbonate rock. (Analysis was performed by Weatherford Laboratories.)
Works referenced in this record:
A new optimization approach to energy network modeling: anthropogenic CO 2 capture coupled with enhanced oil recovery : A new optimization approach to energy network modeling
journal, December 2012
- Middleton, Richard S.
- International Journal of Energy Research, Vol. 37, Issue 14
Coupled alkali-feldspar dissolution and secondary mineral precipitation in batch systems: 1. New experiments at 200 °C and 300 bars
journal, January 2009
- Fu, Qi; Lu, Peng; Konishi, Hiromi
- Chemical Geology, Vol. 258, Issue 3-4
US Department of Energy’s regional carbon sequestration partnership initiative: Update on validation and development phases
journal, January 2011
- Rodosta, Traci; Litynski, John; Plasynski, Sean
- Energy Procedia, Vol. 4
The conditions limiting CO2 storage in aquifers
journal, September 1993
- van der Meer, L. G. H.
- Energy Conversion and Management, Vol. 34, Issue 9-11
Geochemistry of CO2 sequestration in the Jurassic Navajo Sandstone, Colorado Plateau, Utah
journal, February 2007
- Parry, W. T.; Forster, Craig B.; Evans, J. P.
- Environmental Geosciences, Vol. 14, Issue 2
Evaporite Caprock Integrity: An Experimental Study of Reactive Mineralogy and Pore-Scale Heterogeneity during Brine-CO 2 Exposure
journal, August 2012
- Smith, Megan M.; Sholokhova, Yelena; Hao, Yue
- Environmental Science & Technology, Vol. 47, Issue 1
CO2–rock–brine interactions in Lower Tuscaloosa Formation at Cranfield CO2 sequestration site, Mississippi, U.S.A.
journal, January 2012
- Lu, Jiemin; Kharaka, Yousif K.; Thordsen, James J.
- Chemical Geology, Vol. 291
Influence of carbon dioxide on coal permeability determined by pressure transient methods
journal, January 2009
- Siriwardane, Hema; Haljasmaa, Igor; McLendon, Robert
- International Journal of Coal Geology, Vol. 77, Issue 1-2
An experimental investigation of trace element dissolution in carbon dioxide: Applications to the geological storage of CO2
journal, October 2011
- Rempel, Kirsten U.; Liebscher, Axel; Heinrich, Wilhelm
- Chemical Geology, Vol. 289, Issue 3-4
A study of safe CO 2 storage capacity in saline aquifers: a numerical study : CO
journal, October 2012
- Ranjith, P. G.; Perera, M. S. A.; Khan, E.
- International Journal of Energy Research, Vol. 37, Issue 3
CO2–brine–rock interaction — First results of long-term exposure experiments at in situ P–T conditions of the Ketzin CO2 reservoir
journal, August 2010
- Fischer, Sebastian; Liebscher, Axel; Wandrey, Maren
- Geochemistry, Vol. 70
Geochemical effects of CO2 sequestration in sandstones under simulated in situ conditions of deep saline aquifers
journal, September 2008
- Wigand, M.; Carey, J. W.; Schütt, H.
- Applied Geochemistry, Vol. 23, Issue 9
Experimental determination of porosity and permeability changes induced by injection of CO2 into carbonate rocks
journal, July 2009
- Luquot, L.; Gouze, P.
- Chemical Geology, Vol. 265, Issue 1-2
Physical and chemical characteristics of potential seal strata in regions considered for demonstrating geological saline CO2 sequestration
journal, February 2011
- Griffith, Craig A.; Dzombak, David A.; Lowry, Gregory V.
- Environmental Earth Sciences, Vol. 64, Issue 4
X-ray microtomography characterization of porosity, permeability and reactive surface changes during dissolution
journal, March 2011
- Gouze, Philippe; Luquot, Linda
- Journal of Contaminant Hydrology, Vol. 120-121
Reactivity of Mount Simon Sandstone and the Eau Claire Shale Under CO 2 Storage Conditions
journal, August 2012
- Carroll, Susan A.; McNab, Walt W.; Dai, Zurong
- Environmental Science & Technology, Vol. 47, Issue 1
Understanding the significance of in situ coal properties for CO 2 sequestration: An experimental and numerical study : Coal properties for CO2 sequestration
journal, July 2013
- De Silva, P. N. K.; Ranjith, P. G.
- International Journal of Energy Research, Vol. 38, Issue 1
Numerical simulation of porosity and permeability evolution of Mount Simon sandstone under geological carbon sequestration conditions
journal, May 2015
- Zhang, Liwei; Soong, Yee; Dilmore, Robert
- Chemical Geology, Vol. 403
Effect of contaminants from flue gas on CO 2 sequestration in saline formation : CO2 sequestration
journal, December 2013
- Soong, Yee; Hedges, Sheila W.; Howard, Bret H.
- International Journal of Energy Research, Vol. 38, Issue 9
The dissolution kinetics of major sedimentary carbonate minerals
journal, July 2002
- Morse, John W.; Arvidson, Rolf S.
- Earth-Science Reviews, Vol. 58, Issue 1-2
North Dakota lignite and Pittsburgh bituminous coal: a comparative analysis in application to CO<SUB align=right>2 sequestration
journal, January 2011
- Haljasmaa, Igor V.; McLendon, T. Robert; Jikich, Sinisha A.
- International Journal of Oil, Gas and Coal Technology, Vol. 4, Issue 3
Modeling and simulation of carbon sequestration at Cranfield incorporating new physical models
journal, October 2013
- Delshad, Mojdeh; Kong, Xianhui; Tavakoli, Reza
- International Journal of Greenhouse Gas Control, Vol. 18
CO2 Sequestration in Saline Formation
journal, January 2014
- Soong, Yee; Howard, Bret H.; Hedges, Sheila W.
- Aerosol and Air Quality Research, Vol. 14, Issue 2
Works referencing / citing this record:
Application of a new reduced-complexity assessment tool to estimate CO 2 and brine leakage from reservoir and above-zone monitoring interval (AZMI) through an abandoned well under geologic carbon storage conditions : Modeling and Analysis: Application of a new reduced-complexity assessment tool to estimate CO
journal, September 2018
- Zhang, Liwei; Dilmore, Robert; Huerta, Nicolas
- Greenhouse Gases: Science and Technology, Vol. 8, Issue 5
Water–rock–CO 2 interactions and CO 2 storage of Honghe tight oil reservoirs: an experimental and simulation study
journal, May 2019
- He, Yingfu; Ji, Bingyu; Yang, Shu
- Greenhouse Gases: Science and Technology, Vol. 9, Issue 4
Micro-CT Characterization of Wellbore Cement Degradation in SO 4 2- –Bearing Brine under Geological CO 2 Storage Environment
journal, November 2019
- Gan, Manguang; Zhang, Liwei; Miao, Xiuxiu
- Geofluids, Vol. 2019

 Search WorldCat to find libraries that may hold this journal
Search WorldCat to find libraries that may hold this journal