Exploration of imatinib and nilotinib-derived templates as the P2-Ligand for HIV-1 protease inhibitors: Design, synthesis, protein X-ray structural studies, and biological evaluation
Journal Article
·
· European Journal of Medicinal Chemistry
- Purdue Univ., West Lafayette, IN (United States)
- Georgia State Univ., Atlanta, GA (United States)
- Kumamoto University (Japan)
- National Center for Global Health and Medicine Research Institute, Tokyo (Japan)
- Kumamoto University (Japan); National Center for Global Health and Medicine Research Institute, Tokyo (Japan); National Institutes of Health (NIH), Bethesda, MD (United States)
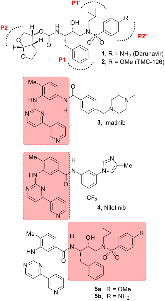
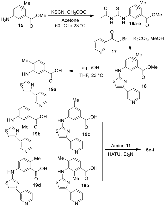
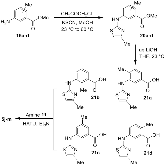
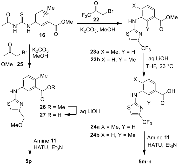
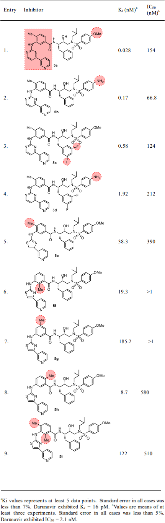
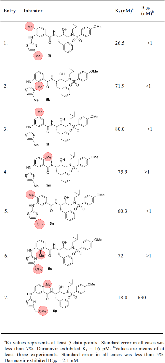
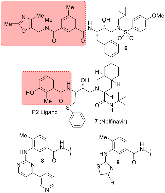
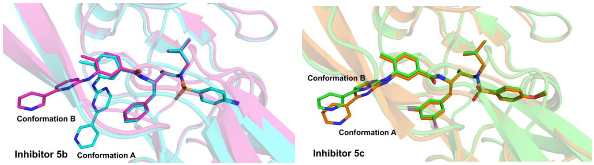
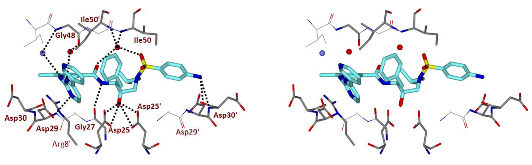
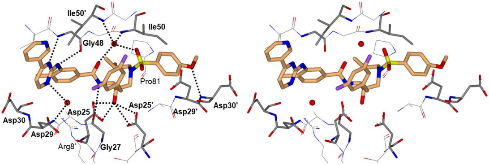
Structure-based design, synthesis, X-ray structural studies, and biological evaluation of a new series of potent HIV-1 protease inhibitors are described. Here, these inhibitors contain various pyridyl-pyrimidine, aryl thiazole or alkylthiazole derivatives as the P2 ligands in combination with darunavir-like hydroxyethylamine sulfonamide isosteres. These heterocyclic ligands are inherent to kinase inhibitor drugs, such as nilotinib and imatinib. These ligands are designed to make hydrogen bonding interactions with the backbone atoms in the S2 subsite of HIV-1 protease. Various benzoic acid derivatives have been synthesized and incorporation of these ligands provided potent inhibitors that exhibited subnanomolar level protease inhibitory activity and low nanomolar level antiviral activity. Two high resolution X-ray structures of inhibitor-bound HIV-1 protease were determined. These structures provided important ligand-binding site interactions for further optimization of this class of protease inhibitors.
- Research Organization:
- Argonne National Laboratory (ANL), Argonne, IL (United States). Advanced Photon Source (APS)
- Sponsoring Organization:
- Japan Agency for Medical Research and Development (AMED); Japan Society for the Promotion of Science (JSPS); National Institutes of Health (NIH); USDOE Office of Science (SC), Basic Energy Sciences (BES)
- Grant/Contract Number:
- AC02-06CH11357; W-31109-ENG-38
- OSTI ID:
- 2423428
- Journal Information:
- European Journal of Medicinal Chemistry, Journal Name: European Journal of Medicinal Chemistry Journal Issue: C Vol. 255; ISSN 0223-5234
- Publisher:
- ElsevierCopyright Statement
- Country of Publication:
- United States
- Language:
- English
Similar Records
Evaluation of darunavir-derived HIV-1 protease inhibitors incorporating P2' amide-derivatives: Synthesis, biological evaluation and structural studies
Design, Synthesis, Biological Evaluation, and X-ray Studies of HIV-1 Protease Inhibitors with Modified P2' Ligands of Darunavir
Design and Development of Highly Potent HIV-1 Protease Inhibitors with a Crown-Like Oxotricyclic Core as the P2-Ligand To Combat Multidrug-Resistant HIV Variants
Journal Article
·
Thu Feb 02 19:00:00 EST 2023
· Bioorganic and Medicinal Chemistry Letters
·
OSTI ID:2423424
Design, Synthesis, Biological Evaluation, and X-ray Studies of HIV-1 Protease Inhibitors with Modified P2' Ligands of Darunavir
Journal Article
·
Sun Nov 05 19:00:00 EST 2017
· ChemMedChem
·
OSTI ID:1422231
Design and Development of Highly Potent HIV-1 Protease Inhibitors with a Crown-Like Oxotricyclic Core as the P2-Ligand To Combat Multidrug-Resistant HIV Variants
Journal Article
·
Mon Apr 17 20:00:00 EDT 2017
· Journal of Medicinal Chemistry
·
OSTI ID:1368286