Calcium-mediated nitrogen reduction for electrochemical ammonia synthesis
Journal Article
·
· Nature Materials
- Technical University of Denmark, Lyngby (Denmark); SLAC
- Stanford University, CA (United States); SLAC National Accelerator Laboratory (SLAC), Menlo Park, CA (United States). SUNCAT Center for Interface Science and Catalysis
- Technical University of Denmark, Lyngby (Denmark)
- SLAC National Accelerator Laboratory (SLAC), Menlo Park, CA (United States). SUNCAT Center for Interface Science and Catalysis
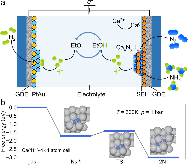
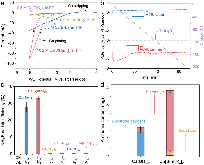
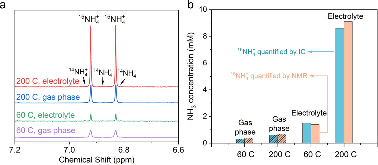
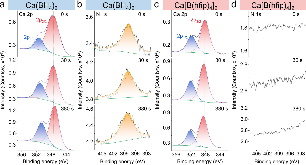
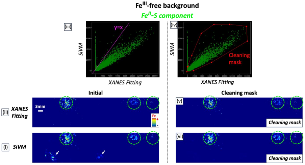
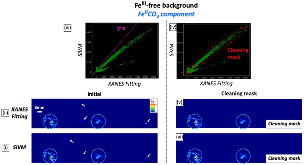
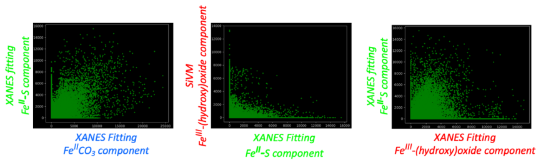
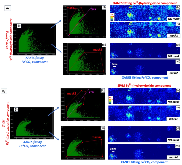
Ammonia (NH3) is a key commodity chemical for the agricultural, textile and pharmaceutical industries, but its production via the Haber–Bosch process is carbon-intensive and centralized. Alternatively, an electrochemical method could enable decentralized, ambient NH3 production that can be paired with renewable energy. The first verified electrochemical method for NH3 synthesis was a process mediated by lithium (Li) in organic electrolytes. So far, however, elements other than Li remain unexplored in this process for potential benefits in efficiency, reaction rates, device design, abundance and stability. In our demonstration of a Li-free system, we found that calcium can mediate the reduction of nitrogen for NH3 synthesis. Here we verified the calcium-mediated process using a rigorous protocol and achieved an NH3 Faradaic efficiency of 40 ± 2% using calcium tetrakis(hexafluoroisopropyloxy)borate (Ca[B(hfip)4]2) as the electrolyte. Our results offer the possibility of using abundant materials for the electrochemical production of NH3, a critical chemical precursor and promising energy vector
- Research Organization:
- SLAC National Accelerator Laboratory (SLAC), Menlo Park, CA (United States)
- Sponsoring Organization:
- Camille and Henry Dreyfus Foundation; European Research Council (ERC); National Science Foundation (NSF); USDOE Office of Science (SC), Basic Energy Sciences (BES). Chemical Sciences, Geosciences & Biosciences Division (CSGB); Villum Fonden
- Grant/Contract Number:
- AC02-76SF00515
- OSTI ID:
- 2310365
- Journal Information:
- Nature Materials, Journal Name: Nature Materials Journal Issue: 1 Vol. 23; ISSN 1476-1122
- Publisher:
- Springer NatureCopyright Statement
- Country of Publication:
- United States
- Language:
- English
Similar Records
Ambient Electrosynthesis of Ammonia: Electrode Porosity and Composition Engineering
Investigation of Rechargeable Calcium Metal-Selenium Batteries Enabled by Borate-Based Electrolytes
Journal Article
·
Tue Jun 19 20:00:00 EDT 2018
· Angewandte Chemie (International Edition)
·
OSTI ID:1476757
Investigation of Rechargeable Calcium Metal-Selenium Batteries Enabled by Borate-Based Electrolytes
Journal Article
·
Tue Mar 07 19:00:00 EST 2023
· Chemistry of Materials
·
OSTI ID:1989122