Elucidating the Mechanism for the Reaction of $$o$$-Phthalaldehyde with Primary Amines in the Presence of Thiols
Journal Article
·
· Journal of Physical Chemistry. B
- Lawrence Berkeley National Laboratory (LBNL), Berkeley, CA (United States)
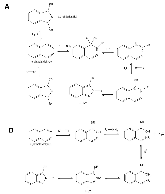
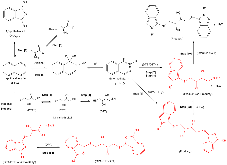
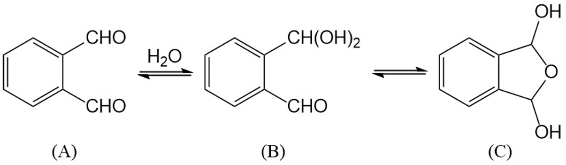
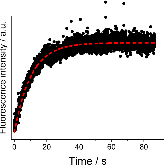
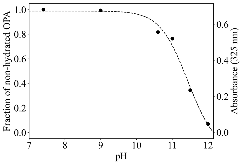
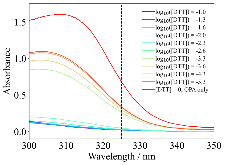
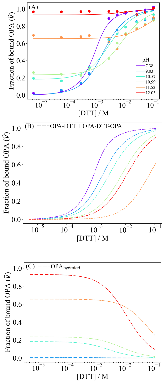
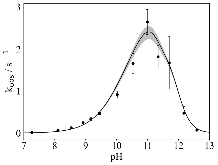
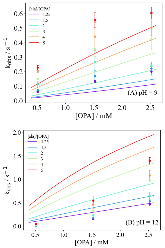
The use of o-phthalaldehyde (OPA) in combination with a thiol reagent is a common method for detecting primary amines in amino acids, peptides, and proteins. Despite its widespread use, the exact reaction mechanism has been debated since the 1980s. Here, we measure the kinetics of the reaction between OPA, alanine, and a dithiol (1,4-dithiolthreitol, DTT) as a function of pH and reagent concentration. Using these new measurements and accompanying kinetic models, we find evidence that the pH dependence of the kinetics arises from both the protonation states of alanine and DTT, the hydration state of OPA, and the unproductive equilibrium with DTT, all of which are pH-dependent. Importantly, these results support the mechanism originally proposed by Sternson [Rational design and evaluation of improved o-phthalaldehyde-like fluorogenic reagents. Anal. Biochem. 1985, 144, 233–246] and Wong [Reaction of o-phthalaldehyde with alanine and thiols: kinetics and mechanism. J. Am. Chem. Soc. 1985, 107, 6421–6422], in which the primary amine first reacts with OPA, followed by a reaction with the thiol to form the fluorescent isoindole product.
- Research Organization:
- Lawrence Berkeley National Laboratory (LBNL), Berkeley, CA (United States)
- Sponsoring Organization:
- USDOE Office of Science (SC), Basic Energy Sciences (BES). Chemical Sciences, Geosciences & Biosciences Division (CSGB)
- Grant/Contract Number:
- AC02-05CH11231
- OSTI ID:
- 2293514
- Journal Information:
- Journal of Physical Chemistry. B, Journal Name: Journal of Physical Chemistry. B Journal Issue: 14 Vol. 127; ISSN 1520-6106
- Publisher:
- American Chemical SocietyCopyright Statement
- Country of Publication:
- United States
- Language:
- English
Similar Records
Fluorescence reagents for high-sensitivity chromatographic measurements of primary amines
Journal Article
·
Thu Sep 01 00:00:00 EDT 1988
· Anal. Chem.; (United States)
·
OSTI ID:7014036