Evidence for distinct rate-limiting steps in the cleavage of alkenes by carotenoid cleavage dioxygenases
Journal Article
·
· Journal of Biological Chemistry
- Case Western Reserve Univ., Cleveland, OH (United States)
- Brookhaven National Lab. (BNL), Upton, NY (United States); Case Western Reserve Univ., Cleveland, OH (United States)
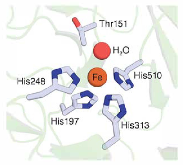
Carotenoid cleavage dioxygenases (CCDs) use a nonheme Fe(II) cofactor to split alkene bonds of carotenoid and stilbenoid substrates. The iron centers of CCDs are typically five-coordinate in their resting states, with solvent occupying an exchangeable site. The involvement of this iron-bound solvent in CCD catalysis has not been experimentally addressed, but computational studies suggest two possible roles. 1) Solvent dissociation provides a coordination site for O2, or 2) solvent remains bound to iron but changes its equilibrium position to allow O2 binding and potentially acts as a proton source. To test these predictions, we investigated isotope effects (H2O versus D2O) on two stilbenoid-cleaving CCDs, Novosphingobium aromaticivorans oxygenase 2 (NOV2) and Neurospora crassa carotenoid oxygenase 1 (CAO1), using piceatannol as a substrate. NOV2 exhibited an inverse isotope effect (kH/kD ~ 0.6) in an air-saturated buffer, suggesting that solvent dissociates from iron during the catalytic cycle. By contrast, CAO1 displayed a normal isotope effect (kH/kD ~ 1.7), suggesting proton transfer in the rate-limiting step. X-ray absorption spectroscopy on NOV2 and CAO1 indicated that the protonation states of the iron ligands are unchanged within pH 6.5–8.5 and that the Fe(II)–aquo bond is minimally altered by substrate binding. In this study, we pinpointed the origin of the differential kinetic behaviors of NOV2 and CAO1 to a single amino acid difference near the solvent-binding site of iron, and X-ray crystallography revealed that the substitution alters binding of diffusible ligands to the iron center. We conclude that solvent-iron dissociation and proton transfer are both associated with the CCD catalytic mechanism.
- Research Organization:
- Brookhaven National Laboratory (BNL), Upton, NY (United States)
- Sponsoring Organization:
- National Institutes of Health (NIH); USDOE Office of Science (SC), Basic Energy Sciences (BES) (SC-22); USDOE Office of Science (SC), Biological and Environmental Research (BER)
- Grant/Contract Number:
- AC02-06CH11357; AC02-76SF00515; SC0012704
- OSTI ID:
- 1679969
- Alternate ID(s):
- OSTI ID: 1557286
- Report Number(s):
- BNL-219982-2020--JAAM
- Journal Information:
- Journal of Biological Chemistry, Journal Name: Journal of Biological Chemistry Journal Issue: 27 Vol. 294; ISSN 0021-9258
- Publisher:
- American Society for Biochemistry and Molecular BiologyCopyright Statement
- Country of Publication:
- United States
- Language:
- English
A modular pathway engineering strategy for the high-level production of β-ionone in Yarrowia lipolytica
|
journal | February 2020 |
Similar Records
Spectroscopy and crystallography define carotenoid oxygenases as a new subclass of mononuclear non-heme FeII enzymes
Structure and Spectroscopy of Alkene-Cleaving Dioxygenases Containing an Atypically Coordinated Non-Heme Iron Center
Journal Article
·
Mon Mar 24 20:00:00 EDT 2025
· Journal of Biological Chemistry
·
OSTI ID:2539936
Structure and Spectroscopy of Alkene-Cleaving Dioxygenases Containing an Atypically Coordinated Non-Heme Iron Center
Journal Article
·
Wed May 10 20:00:00 EDT 2017
· Biochemistry
·
OSTI ID:1376251