Enhancing Oxygen Exchange Activity by Tailoring Perovskite Surfaces
- Univ. of Pennsylvania, Philadelphia, PA (United States)
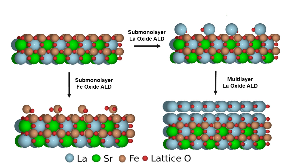
A detailed understanding of the effects of surface chemical and geometric composition is essential for understanding the electrochemical performance of the perovskite (ABO3) oxides commonly used as electrocatalysts in the cathodes of ceramic fuel cells. In this work, we report how the addition of submonolayer quantities of A- and B-site cations affect the rate of the Oxygen Reduction Reaction (ORR) of Sr-doped LaFeO3 (LSF), LaMnO3 (LSM), and LaCoO3 (LSCo). Density functional theory (DFT) calculations were performed to determine the stability of different active sites on a collection of surfaces. With LSF and LSM, rates for ORR are significantly higher on the A-site terminated surface, while surface termination is less important for LSCo. Our findings highlight the importance of tailoring the surface termination of the perovskite to obtain its ultimate ORR performance.
- Research Organization:
- Univ. of Pennsylvania, Philadelphia, PA (United States)
- Sponsoring Organization:
- USDOE Office of Fossil Energy (FE)
- Grant/Contract Number:
- FE0031252
- OSTI ID:
- 1594019
- Journal Information:
- Journal of Physical Chemistry Letters, Vol. 10, Issue 14; ISSN 1948-7185
- Publisher:
- American Chemical SocietyCopyright Statement
- Country of Publication:
- United States
- Language:
- English
Web of Science
Similar Records
CH4 Activation over Perovskite Catalysts: True Density and Reactivity of Active Sites
The role of nonmetallic ion substitution in perovskite LaCoO3 for improved oxygen evolution reaction activity