Mo6S8-based single-metal-atom catalysts for direct methane to methanol conversion
Journal Article
·
· Journal of Chemical Physics
- Michigan Technological Univ., Houghton, MI (United States)
- Yangzhou Univ. (China)
- Brookhaven National Lab. (BNL), Upton, NY (United States)
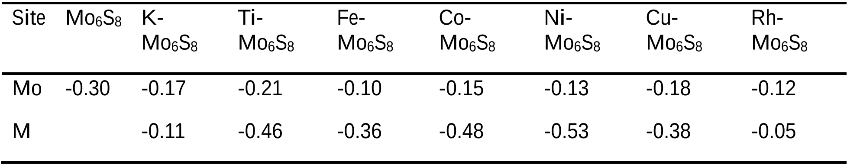
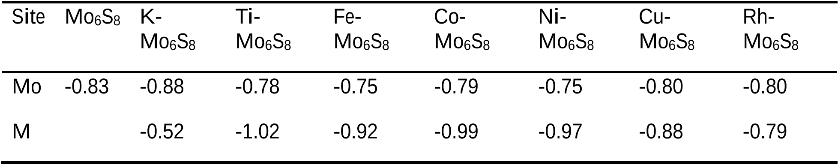
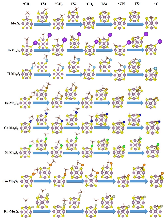
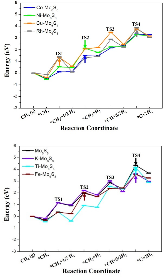
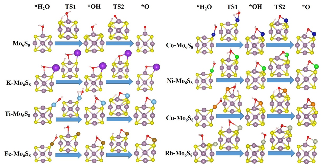
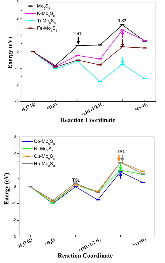
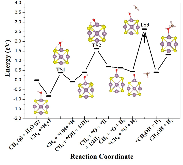
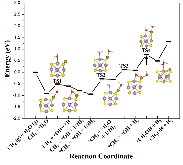
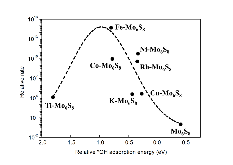
The single atom catalysts have been attracting much attention for catalysis. Here, the significant influence of single-metal-atom (M = K, Ti, Fe, Co, Ni, Cu, Rh) doping on a Mo6S8 cluster was revealed for the direct methane to methanol conversion in water stream using density functional theory calculations. It was found that all single atom dopants help to facilitate the conversion via the steam reforming of methane (SRM). The single Fe atom on Mo6S8 (Fe—Mo6S8) exhibits the most significant promoting effect, which is followed by Ni, Co, Rh—Mo6S8 > K, Ti, Cu—Mo6S8 > Mo6S8 in a decreasing sequence. The enhanced activity by single atom doping on Mo6S8 is mainly associated with the interplay between the ensemble effect via the direct participation of an active M dopant and the site confinement imposed by doping of a single M atom, in tuning the methane conversion and methanol selectivity. It generates the new active center, M, which confines the SRM to occur at the M—Mo bridge sites and facilitates the selective production of methanol. An adequate single-atom promoter should not only bind *OH or *O moderately, being strongly enough to help water dissociation and weakly enough to allow the oxidation of methane, but also impose the confinement effect to facilitate the C—O bond association and production of methanol. Our results highlight the importance of the interplay among ligand, ensemble, and confinement effects in promoting the complex SRM over single atom catalysts.
- Research Organization:
- Brookhaven National Laboratory (BNL), Upton, NY (United States)
- Sponsoring Organization:
- USDOE; USDOE Office of Science (SC), Basic Energy Sciences (BES) (SC-22). Chemical Sciences, Geosciences & Biosciences Division
- Grant/Contract Number:
- SC0012704
- OSTI ID:
- 1530524
- Alternate ID(s):
- OSTI ID: 1568928
- Report Number(s):
- BNL--211823-2019-JAAM
- Journal Information:
- Journal of Chemical Physics, Journal Name: Journal of Chemical Physics Journal Issue: 2 Vol. 151; ISSN 0021-9606
- Publisher:
- American Institute of Physics (AIP)Copyright Statement
- Country of Publication:
- United States
- Language:
- English
Solar Driven CO2 Hydrogenation on Ti-Doped Silicon Nanocages
|
journal | November 2019 |
Similar Records
Mechanistic study of methanol synthesis from CO₂ and H₂ on a modified model Mo₆S₈ cluster
Direct Oxidation of Methane to Methanol Enabled by Electronic Atomic Monolayer–Metal Support Interaction
Journal Article
·
Sun Jan 11 19:00:00 EST 2015
· ACS Catalysis
·
OSTI ID:1183833
Direct Oxidation of Methane to Methanol Enabled by Electronic Atomic Monolayer–Metal Support Interaction
Journal Article
·
Sun Jun 02 20:00:00 EDT 2019
· ACS Catalysis
·
OSTI ID:1577539