Copper/TEMPO Redox Redux: Analysis of PCET Oxidation of TEMPOH by Copper(II) and the Reaction of TEMPO with Copper(I)
- Univ. of Wisconsin, Madison, WI (United States)
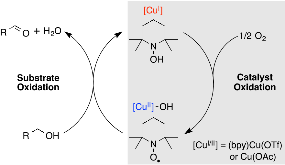
Copper salts and organic aminoxyls, such as TEMPO (2,2,6,6- tetramethylpiperidine N-oxyl), are versatile catalysts for aerobic alcohol oxidation. Previous reports in the literature contain conflicting proposals concerning the redox interactions that take place between copper(I) and copper(II) salts with the aminoxyl and hydroxylamine species, TEMPO and TEMPOH, respectively. In this work, we reinvestigate these reactions in an effort to resolve the conflicting claims in the literature. Under anaerobic conditions, CuIIX2 salts [X= acetate (OAc), trifluoroacetate (TFA), triflate (OTf)] are shown to promote rapid proton-coupled oxidation of TEMPOH to TEMPO: CuIIX2 + TEMPOH → CuIX + TEMPO + HX. In the reaction with acetate, yet, slow reoxidation of CuIOAc occurs. This process requires both TEMPO and HOAc and coincides with reduction of TEMPO to 2,2,6,6-tetramethylpiperidine. Analogous reactivity is not observed with trifluoroacetate and triflate species. As a whole, the facility of proton-coupled oxidation of TEMPOH by CuII salts suggests that this process could contribute to catalyst regeneration under aerobic oxidation conditions.
- Research Organization:
- Univ. of Wisconsin, Madison, WI (United States)
- Sponsoring Organization:
- USDOE Office of Science (SC), Basic Energy Sciences (BES); National Institutes of Health (NIH); National Science Foundation (NSF)
- Grant/Contract Number:
- FG02-05ER15690; CHE-1048642; CHE-0741901
- OSTI ID:
- 1544423
- Journal Information:
- Inorganic Chemistry, Vol. 58, Issue 15; Related Information: Supporting Information is available free of charge on the ACS Publications website at DOI: 10.1021/acs.inorgchem.9b01326.; ISSN 0020-1669
- Publisher:
- American Chemical Society (ACS)Copyright Statement
- Country of Publication:
- United States
- Language:
- English
Web of Science
Similar Records
Electrochemical Oxidation of Organic Molecules at Lower Overpotential: Accessing Broader Functional Group Compatibility with Electron–Proton Transfer Mediators
Electrochemical Oxidation of Organic Molecules at Lower Overpotential: Accessing Broader Functional Group Compatibility with Electron-Proton Transfer Mediators