Tuning phase stability and short-range order through Al doping in high-entropy alloys
- Ames Lab., Ames, IA (United States)
- RWTH Aachen Univ. (Germany)
- Lehigh Univ., Bethlehem, PA (United States)
- RWTH Aachen Univ. (Germany); Indian Inst. of Technology (IIT), Madras (India)
- Ames Lab. and Iowa State Univ., Ames, IA (United States)
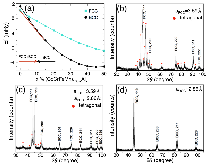
For high-entropy alloys, we report on the phase evolution with increasing Al content ( 0 ≤ x ≤ 20 at.%). From first-principles theory, aluminum doping drives the alloy structurally from fcc to bcc separated by a narrow two-phase region (fcc+bcc), which is well supported by our experiments. Using KKR-CPA electronic-structure calculations, we highlight the effect of Al doping on the formation enthalpy (alloy stability) and electronic dispersion of alloys. As chemical short-range order indicates the nascent local order, and entropy changes, as well as expected low-temperature ordering behavior, we use KKR-CPA-based thermodynamic linear response to predict the chemical ordering behavior of arbitrary complex solid-solution alloys—an ideal approach for predictive design of high-entropy alloys. The predictions confirm our present experimental findings and other reported ones.
- Research Organization:
- Ames Laboratory (AMES), Ames, IA (United States)
- Sponsoring Organization:
- USDOE Office of Science (SC), Basic Energy Sciences (BES) (SC-22). Materials Sciences & Engineering Division; USDOE Office of Science (SC), Basic Energy Sciences (BES)
- Grant/Contract Number:
- AC02-07CH11358; N00014-16-1-2548
- OSTI ID:
- 1542874
- Alternate ID(s):
- OSTI ID: 1546452
- Report Number(s):
- IS-J-9970; PRMHAR
- Journal Information:
- Physical Review Materials, Vol. 3, Issue 7; ISSN 2475-9953
- Publisher:
- American Physical Society (APS)Copyright Statement
- Country of Publication:
- United States
- Language:
- English
Web of Science
Effect of Solution Treatment on the Shape Memory Functions of (TiZrHf)50Ni25Co10Cu15 High Entropy Shape Memory Alloy
|
journal | October 2019 |
Similar Records
Towards cubic symmetry for : Structure and magnetism of the antifluorite
Magnetic and magnetocaloric properties of compounds (x = 0.25, 0.5, 0.75)