Reversible anionic redox activity in Na3RuO4 cathodes: a prototype Na-rich layered oxide
Journal Article
·
· Energy & Environmental Science
- National Inst. of Advanced Industrial Science and Technology (AIST), Tsukuba (Japan). Energy Technology Research Inst.; Univ. of Tsukuba (Japan). Graduate School of System and Information Engineering
- Nanjing Univ. (China). Center of Energy Storage Materials & Technology. College of Engineering and Applied Sciences. National Lab. of Solid State Microstructures. Collaborative Innovation Center of Advanced Microstructures
- National Inst. of Advanced Industrial Science and Technology (AIST), Tsukuba (Japan). Energy Technology Research Inst.
- Shanghai Jiao Tong Univ. (China). State Key Lab. of Metal Matrix Composites. School of Material Science and Engineering
- Argonne National Lab. (ANL), Argonne, IL (United States). X-ray Science Division
- Nanjing Univ. (China). Center of Energy Storage Materials & Technology. College of Engineering and Applied Sciences. National Lab. of Solid State Microstructures. Collaborative Innovation Center of Advanced Microstructures; National Inst. of Advanced Industrial Science and Technology (AIST), Tsukuba (Japan). Energy Technology Research Inst.; Univ. of Tsukuba (Japan). Graduate School of System and Information Engineering
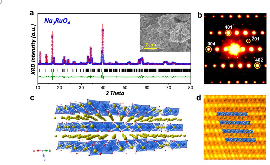
Sodium-ion batteries are attractive for large-scale energy storage due to the abundance of sodium, but the deficient capacity achieved by cathode materials prevents their further applications. Chemical substitution of Na in transition metal layers is a promising solution to utilize both the cationic and anionic redox activities for boosting energy storage. Unfortunately, different from the classic Li-rich Li2MnO3, a pure prototype with anionic redox activity has not been found among the typical Na-rich cathodes. In this paper, we originally design a Na-rich layered oxide prototype, namely Na3RuO4 (Ru5+), which delivers a partial reversible capacity solely via the participation of oxygen anions. More importantly, the anionic redox activity is validated by the in situ Raman observation of reversible peroxo-based O–O (de)bonding upon cycling. Finally, our findings not only highlight the multiple electron-transfer strategy for capacity extension, but also broaden the horizon in designing Na-rich electrode materials for high-energy sodium-ion batteries.
- Research Organization:
- Argonne National Lab. (ANL), Argonne, IL (United States); Nanjing Univ. (China); National Inst. of Advanced Industrial Science and Technology (AIST), Tsukuba (Japan); Shanghai Jiao Tong Univ. (China)
- Sponsoring Organization:
- China Scholarship Council (CSC); Japan Science and Technology Agency (JST); National Basic Research Program of China; National Natural Science Foundation of China (NSFC); Natural Science Foundation of Jiangsu Province (China); USDOE Office of Science (SC)
- Grant/Contract Number:
- AC02-06CH11357
- OSTI ID:
- 1471523
- Journal Information:
- Energy & Environmental Science, Journal Name: Energy & Environmental Science Journal Issue: 2 Vol. 11; ISSN 1754-5692
- Publisher:
- Royal Society of ChemistryCopyright Statement
- Country of Publication:
- United States
- Language:
- English
Similar Records
Quantification of Anionic Redox Chemistry in a Prototype Na-Rich Layered Oxide
Journal Article
·
Sun Dec 29 19:00:00 EST 2019
· ACS Applied Materials and Interfaces
·
OSTI ID:1780734