Downregulation of p-COUMAROYL ESTER 3-HYDROXYLASE in rice leads to altered cell wall structures and improves biomass saccharification
- Kyoto Univ. (Japan). Research Inst. for Sustainable Humanosphere; Research Institute for Sustainable Humanosphere, Kyoto University
- Kyoto Univ. (Japan). Research Inst. for Sustainable Humanosphere
- Univ. of Wisconsin, Madison, WI (United States); Great Lakes Bioenergy Research Center (GLBRC), Madison, WI (United States)
- Tokushima Univ., Kuramoto-cho, Tokushima (Japan). Faculty of Bioscience and Bioindustry
- Kyoto Univ. (Japan). Graduate School of Agriculture
- Kyoto Univ. (Japan). Research Inst. for Sustainable Humanosphere; Kyoto Univ. (Japan). Research Unit for Development of Global Sustainability
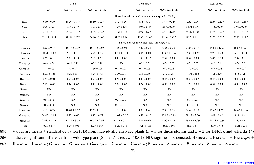
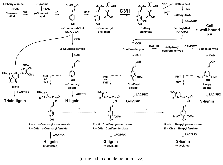
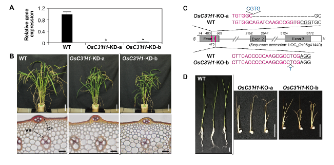
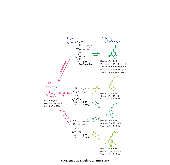
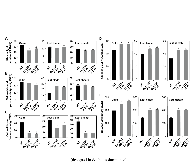
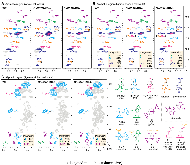
p-Coumaroyl ester 3-hydroxylase (C3'H) is a key enzyme involved in the biosynthesis of lignin, a phenylpropanoid polymer that is the major constituent in vascular plants’ secondary cell walls. Although the crucial role of C3'H in lignification and its manipulation to upgrade lignocellulose have been investigated in eudicots, limited information is available in monocotyledonous grass species despite their potential as biomass feedstocks. Here in this paper we address the pronounced impacts of C3'H-deficiency on the structure and properties of grass cell walls. C3'H-knockdown lines generated via RNAi-mediated gene silencing, with ~0.5% of residual expression levels, reached maturity and set seeds, whereas, in contrast, C3'H-knockout rice mutants generated via CRISPR/Cas9-mediated mutagenesis were severely dwarfed and sterile. Cell wall analysis of the mature C3'H-knockdown RNAi lines revealed that their lignins were largely enriched in p-hydroxyphenyl (H) units while substantially reduced in the normally dominant guaiacyl (G) and syringyl (S) units. Interestingly, however, the enrichment of H units was limited to being within the non-acylated lignin units, with grass-specific γ-p-coumaroylated lignin units remaining apparently unchanged. Suppression of C3'H also resulted in relative augmentation in tricin residues in lignin as well as substantial reduction in wall cross-linking ferulates. Collectively, our data demonstrate that C3'H expression is an important determinant, not only of lignin content and composition, but also of the degree of cell wall cross-linking. We also demonstrated that C3'H-suppressed rice displays enhanced biomass saccharification.
- Research Organization:
- Great Lakes Bioenergy Research Center (GLBRC), Madison, WI (United States); Univ. of Wisconsin, Madison, WI (United States)
- Sponsoring Organization:
- Japan International Cooperation Agency; Japan Science and Technology Agency (JST); Japan Society for the Promotion of Science (JSPS); Ministry of Agriculture, Forestry and Fisheries of Japan; New Energy and Industrial Technology Development Organization (NEDO); USDOE; USDOE Office of Science (SC), Biological and Environmental Research (BER)
- Grant/Contract Number:
- FC02-07ER64494; SC0018409
- OSTI ID:
- 1459572
- Alternate ID(s):
- OSTI ID: 1459711
- Journal Information:
- The Plant Journal, Journal Name: The Plant Journal Journal Issue: 5 Vol. 95; ISSN 0960-7412
- Publisher:
- Society for Experimental BiologyCopyright Statement
- Country of Publication:
- United States
- Language:
- English
Similar Records
Disruption of p-coumaroyl-CoA:monolignol transferases in rice drastically alters lignin composition
OsCAldOMT1 is a bifunctional O-methyltransferase involved in the biosynthesis of tricin-lignins in rice cell walls
Journal Article
·
Thu Oct 12 20:00:00 EDT 2023
· Plant Physiology (Bethesda)
·
OSTI ID:2294081
OsCAldOMT1 is a bifunctional O-methyltransferase involved in the biosynthesis of tricin-lignins in rice cell walls
Journal Article
·
Sun Aug 11 20:00:00 EDT 2019
· Scientific Reports
·
OSTI ID:1619549