Biomarkers of Tuberculosis Severity and Treatment Effect: A Directed Screen of 70 Host Markers in a Randomized Clinical Trial
- Meso Scale Diagnostics, LLC, Rockville, MD (United States)
- Univ. of California, San Francisco, CA (United States)
- Yale Univ., New Haven, CT (United States). School of Public Health and School of Medicine
- San Antonio VA Medical Center, TX (United States)
- Centers for Disease Control and Prevention (CDC), Atlanta, GA (United States)
- Pacific Northwest National Lab. (PNNL), Richland, WA (United States)
- Univ. of Notre Dame, IN (United States)
- Oregon Health and Science Univ., Portland, OR (United States)
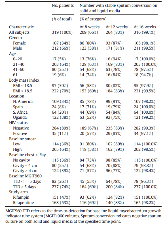
More efficacious treatment regimens are needed for tuberculosis, however, drug development is impeded by a lack of reliable biomarkers of disease severity and of treatment effect. In this study, we conducted a directed screen of host biomarkers in participants enrolled in a tuberculosis clinical trial to address this need. Serum samples from 319 protocol-correct, culture-confirmed pulmonary tuberculosis patients treated under direct observation as part of an international, phase 2 trial were screened for 70 markers of infection, inflammation, and metabolism. Biomarker assays were specifically developed for this study and quantified using a novel, multiplexed electrochemiluminescence assay. We evaluated the association of biomarkers with baseline characteristics, as well as with detailed microbiologic data, using Bonferroni-adjusted, linear regression models. Across numerous analyses, seven proteins, SAA1, PCT, IL-1, IL-6, CRP, PTX-3 and MMP-8, showed recurring strong associations with markers of baseline disease severity, smear grade and cavitation; were strongly modulated by tuberculosis treatment; and had responses that were greater for patients who culture-converted at 8 weeks. With treatment, all proteins decreased, except for osteocalcin, MCP-1 and MCP-4, which significantly increased. Several previously reported putative tuberculosis-associated biomarkers (HOMX1, neopterin, and cathelicidin) were not significantly associated with treatment response. In conclusion, across a geographically diverse and large population of tuberculosis patients enrolled in a clinical trial, several previously reported putative biomarkers were not significantly associated with treatment response, however, seven proteins had recurring strong associations with baseline radiographic and microbiologic measures of disease severity, as well as with early treatment response, deserving additional study.
- Research Organization:
- Pacific Northwest National Laboratory (PNNL), Richland, WA (United States)
- Sponsoring Organization:
- USDOE; National Institutes of Health (NIH); National Institute of Allergy and Infectious Diseases (NIAID); TB Surrogate Markers for Assessing Response to Treatment (TB SMART Study); Centers for Disease Control and Prevention (CDC), TB Trials Consortium
- Grant/Contract Number:
- AC05-76RL01830; 200-2009-32597
- OSTI ID:
- 1439710
- Report Number(s):
- PNNL-SA-129994; 49581; 453060036
- Journal Information:
- EBioMedicine, Vol. 25, Issue C; ISSN 2352-3964
- Publisher:
- ElsevierCopyright Statement
- Country of Publication:
- United States
- Language:
- English
Web of Science
Similar Records
Application of multiplexed ion mobility spectrometry towards the identification of host protein signatures of treatment effect in pulmonary tuberculosis
Nutritional markers and proteome in patients undergoing treatment for pulmonary tuberculosis differ by geographic region