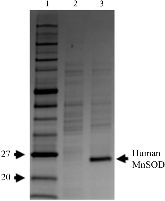
Preliminary neutron diffraction analysis of challenging human manganese superoxide dismutase crystals
Superoxide dismutases (SODs) are enzymes that protect against oxidative stress by dismutation of superoxide into oxygen and hydrogen peroxide through cyclic reduction and oxidation of the active-site metal. The complete enzymatic mechanisms of SODs are unknown since data on the positions of hydrogen are limited. Here, methods are presented for large crystal growth and neutron data collection of human manganese SOD (MnSOD) using perdeuteration and the MaNDi beamline at Oak Ridge National Laboratory. The crystal from which the human MnSOD data set was obtained is the crystal with the largest unit-cell edge (240 Å) from which data have been collected via neutron diffraction to sufficient resolution (2.30 Å) where hydrogen positions can be observed.
- Research Organization:
- Oak Ridge National Laboratory (ORNL), Oak Ridge, TN (United States)
- Sponsoring Organization:
- USDOE Office of Science (SC), Basic Energy Sciences (BES); National Aeronautics and Space Administration (NASA); National Institutes of Health (NIH)
- Grant/Contract Number:
- AC05-00OR22725; 44-0307-1021-201; P30CA036727; 5P20RR016469
- OSTI ID:
- 1437709
- Alternate ID(s):
- OSTI ID: 1407982
- Journal Information:
- Acta Crystallographica. Section F, Structural Biology Communications, Journal Name: Acta Crystallographica. Section F, Structural Biology Communications Vol. 73 Journal Issue: 4; ISSN 2053-230X
- Publisher:
- International Union of Crystallography (IUCr)Copyright Statement
- Country of Publication:
- United Kingdom
- Language:
- English
Web of Science
Similar Records
Redox manipulation of the manganese metal in human manganese superoxide dismutase for neutron diffraction
Direct detection of coupled proton and electron transfers in human manganese superoxide dismutase