The use of a mercury biosensor to evaluate the bioavailability of mercury-thiol complexes and mechanisms of mercury uptake in bacteria
- Rutgers Univ., New Brunswick, NJ (United States); Duke Univ., Durham, NC (United States)
- Rutgers Univ., New Brunswick, NJ (United States)
- Univ. of Connecticut, Groton, CT (United States)
- Harvard School of Public Health, Boston, MA (United States)
- King Abdul-Aziz Univ., Jeddah (Saudi Arabia)
- Cornell Univ., Ithaca, NY (United States)
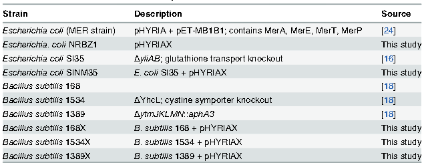
As mercury (Hg) biosensors are sensitive to only intracellular Hg, they are useful in the investigation of Hg uptake mechanisms and the effects of speciation on Hg bioavailability to microbes. In this study, bacterial biosensors were used to evaluate the roles that several transporters such as the glutathione, cystine/cysteine, and Mer transporters play in the uptake of Hg from Hg-thiol complexes by comparing uptake rates in strains with functioning transport systems to strains where these transporters had been knocked out by deletion of key genes. The Hg uptake into the biosensors was quantified based on the intracellular conversion of inorganic mercury (Hg(II)) to elemental mercury (Hg(0)) by the enzyme MerA. It was found that uptake of Hg from Hg-cysteine (Hg(CYS)2) and Hg-glutathione (Hg(GSH)2) complexes occurred at the same rate as that of inorganic complexes of Hg(II) into Escherichia coli strains with and without intact Mer transport systems. However, higher rates of Hg uptake were observed in the strain with a functioning Mer transport system. These results demonstrate that thiol-bound Hg is bioavailable to E. coli and that this bioavailability is higher in Hg-resistant bacteria with a complete Mer system than in non-resistant strains. No difference in the uptake rate of Hg from Hg(GSH)2 was observed in E. coli strains with or without functioning glutathione transport systems. There was also no difference in uptake rates between a wildtype Bacillus subtilis strain with a functioning cystine/cysteine transport system, and a mutant strain where this transport system had been knocked out. These results cast doubt on the viability of the hypothesis that the entire Hg-thiol complex is taken up into the cell by a thiol transporter. It is more likely that the Hg in the Hg-thiol complex is transferred to a transport protein on the cell membrane and is subsequently internalized.
- Research Organization:
- Rutgers Univ., New Brunswick, NJ (United States); Rutgers Univ., Piscataway, NJ (United States)
- Sponsoring Organization:
- USDOE Office of Science (SC), Biological and Environmental Research (BER)
- Grant/Contract Number:
- SC0007051; sc0007051
- OSTI ID:
- 1324975
- Alternate ID(s):
- OSTI ID: 1454700
- Journal Information:
- PLoS ONE, Vol. 10, Issue 9; ISSN 1932-6203
- Publisher:
- Public Library of ScienceCopyright Statement
- Country of Publication:
- United States
- Language:
- English
Web of Science
Similar Records
Direct Measurement of Mercury(II) Removal from Organomercurial Lyase (MerB) by Tryptophan Fluorescence: NmerA Domain of Coevolved γ-Proteobacterial Mercuric Ion Reductase (MerA) Is More Efficient Than MerA Catalytic Core or Glutathione,
Rates and Dynamics of Mercury Isotope Exchange between Dissolved Elemental Hg(0) and Hg(II) Bound to Organic and Inorganic Ligands