Impact of Ligand Substitutions on Multielectron Redox Properties of Fe Complexes Supported by Nitrogenous Chelates
Abstract
Redox flow batteries (RFBs) have recently been recognized as a potentially viable technology for scalable energy storage. To take full advantage of RFBs, one possible approach for achieving high energy densities is to maximize a number of redox events by utilizing charge carriers capable of multiple one-electron transfers within the electrochemical window of solvent. However, past efforts to develop more efficient electrolytes for nonaqueous RFBs have mostly been empirical. In this manuscript, we shed light on design principles by theoretically investigating the effects of systematically substituting pyridyl moieties with imine ligands within a series of Fe complexes with some experimental validation. We found that such replacement is an effective strategy for reducing the molecular weight-to-charge ratios of these complexes. Simultaneously, calculations suggest that the reduction potentials and ligand-based redox activity of such substituted N-heterocyclic Fe compounds might be maintained within their +4 → –1 charge states. Additionally, by theoretically examining the role of coordination geometry, vis-à-vis reducing the number of redox noninnocent ligands within the first coordination sphere, we have demonstrated that Fe complexes with one such ligand were also capable of supporting multielectron reduction events and exhibited reduction potentials similar to their parent analogs supported by two or threemore »
- Authors:
- Publication Date:
- Research Org.:
- Los Alamos National Laboratory (LANL), Los Alamos, NM (United States)
- Sponsoring Org.:
- USDOE Laboratory Directed Research and Development (LDRD) Program
- OSTI Identifier:
- 1480560
- Alternate Identifier(s):
- OSTI ID: 1482944
- Report Number(s):
- LA-UR-18-22442
Journal ID: ISSN 2470-1343
- Grant/Contract Number:
- 20170046DR; 89233218CNA000001
- Resource Type:
- Published Article
- Journal Name:
- ACS Omega
- Additional Journal Information:
- Journal Name: ACS Omega Journal Volume: 3 Journal Issue: 11; Journal ID: ISSN 2470-1343
- Publisher:
- American Chemical Society
- Country of Publication:
- United States
- Language:
- English
- Subject:
- 37 INORGANIC, ORGANIC, PHYSICAL, AND ANALYTICAL CHEMISTRY; Energy Sciences; Inorganic and Physical Chemistry
Citation Formats
Popov, Ivan A., Mehio, Nada, Chu, Terry, Davis, Benjamin L., Mukundan, Rangachary, Yang, Ping, and Batista, Enrique R. Impact of Ligand Substitutions on Multielectron Redox Properties of Fe Complexes Supported by Nitrogenous Chelates. United States: N. p., 2018.
Web. doi:10.1021/acsomega.8b01921.
Popov, Ivan A., Mehio, Nada, Chu, Terry, Davis, Benjamin L., Mukundan, Rangachary, Yang, Ping, & Batista, Enrique R. Impact of Ligand Substitutions on Multielectron Redox Properties of Fe Complexes Supported by Nitrogenous Chelates. United States. https://doi.org/10.1021/acsomega.8b01921
Popov, Ivan A., Mehio, Nada, Chu, Terry, Davis, Benjamin L., Mukundan, Rangachary, Yang, Ping, and Batista, Enrique R. Fri .
"Impact of Ligand Substitutions on Multielectron Redox Properties of Fe Complexes Supported by Nitrogenous Chelates". United States. https://doi.org/10.1021/acsomega.8b01921.
@article{osti_1480560,
title = {Impact of Ligand Substitutions on Multielectron Redox Properties of Fe Complexes Supported by Nitrogenous Chelates},
author = {Popov, Ivan A. and Mehio, Nada and Chu, Terry and Davis, Benjamin L. and Mukundan, Rangachary and Yang, Ping and Batista, Enrique R.},
abstractNote = {Redox flow batteries (RFBs) have recently been recognized as a potentially viable technology for scalable energy storage. To take full advantage of RFBs, one possible approach for achieving high energy densities is to maximize a number of redox events by utilizing charge carriers capable of multiple one-electron transfers within the electrochemical window of solvent. However, past efforts to develop more efficient electrolytes for nonaqueous RFBs have mostly been empirical. In this manuscript, we shed light on design principles by theoretically investigating the effects of systematically substituting pyridyl moieties with imine ligands within a series of Fe complexes with some experimental validation. We found that such replacement is an effective strategy for reducing the molecular weight-to-charge ratios of these complexes. Simultaneously, calculations suggest that the reduction potentials and ligand-based redox activity of such substituted N-heterocyclic Fe compounds might be maintained within their +4 → –1 charge states. Additionally, by theoretically examining the role of coordination geometry, vis-à-vis reducing the number of redox noninnocent ligands within the first coordination sphere, we have demonstrated that Fe complexes with one such ligand were also capable of supporting multielectron reduction events and exhibited reduction potentials similar to their parent analogs supported by two or three of the same multidentate ligands. However, some differences in redox nature within the lower (+2 → –1) charge states were also noticed. Specifically, complexes containing two bidentate ligands, or one tridentate ligand, exhibited ligand-based reductions, whereas compounds with one bidentate ligand exhibited metal-centered reductions. In conclusion, the current results pave the way toward the design of the next-generation of Fe complexes with lower molecular weights and greater stored energy for redox flow batteries.},
doi = {10.1021/acsomega.8b01921},
journal = {ACS Omega},
number = 11,
volume = 3,
place = {United States},
year = {Fri Nov 02 00:00:00 EDT 2018},
month = {Fri Nov 02 00:00:00 EDT 2018}
}
https://doi.org/10.1021/acsomega.8b01921
Web of Science
Figures / Tables:
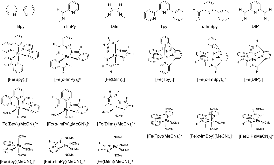 Figure 1: Fe complexes with N-heterocyclic ligands considered in this study, $n$ = +4 → −1.
Figure 1: Fe complexes with N-heterocyclic ligands considered in this study, $n$ = +4 → −1.
Works referencing / citing this record:
Consequences of ligand derivatization on the electronic properties of polyoxovanadate-alkoxide clusters
journal, March 2019
- Schurr, Bradley E.; Nachtigall, Olaf; VanGelder, Lauren E.
- Journal of Coordination Chemistry, Vol. 72, Issue 8
Consequences of ligand derivatization on the electronic properties of polyoxovanadate-alkoxide clusters
text, January 2019
- Schurr, Bradley E.; Nachtigall, Olaf; VanGelder, Lauren E.
- Taylor & Francis
Consequences of ligand derivatization on the electronic properties of polyoxovanadate-alkoxide clusters
journal, March 2019
- Schurr, Bradley E.; Nachtigall, Olaf; VanGelder, Lauren E.
- Journal of Coordination Chemistry, Vol. 72, Issue 8
Consequences of ligand derivatization on the electronic properties of polyoxovanadate-alkoxide clusters
text, January 2019
- Schurr, Bradley E.; Nachtigall, Olaf; VanGelder, Lauren E.
- Taylor & Francis
Figures / Tables found in this record:

 Search WorldCat to find libraries that may hold this journal
Search WorldCat to find libraries that may hold this journal