Electrosorption of organic acids from aqueous bio-oil and conversion into hydrogen via microbial electrolysis cells
Abstract
Neutralization of the bio-oil pH has been shown to generate a neutralized bio-oil aqueous phase (NBOAP) that includes most of the acidic components and a neutralized bio-oil organic phase (NBOOP) that includes hydrophobic organics, such as phenols. NBOOP can be used for fuel production, while NBOAP can be fed to microbial electrolysis cells (MECs) for hydrogen production. After pH neutralization, some organic acidic components remain in NBOOP. This work is focused on capturing acidic compounds from NBOOP through water extraction and electrosorption, and demonstrating hydrogen production via MECs. Capacitive deionization (CDI) is proven effective in capturing ions from NBOOP-contacted water and NBOAP via electrosorption. Captured acidic compounds enable the MEC application to effectively produce renewable hydrogen. Chemical oxygen demand (COD) removal of 49.2%, 61.5%, and 60.8% for 2, 4, and 10 g/L-anode/day loading were observed, corresponding to a total COD degradation of 0.19 g/L, 0.79 g/L, and 1.3 g/L, respectively. A maximum hydrogen productivity of 4.3 L-H2/L-anode/day was obtained. Major compounds in the water phase such as fatty acids, sugar derivatives, furanic and phenolic compounds were converted to hydrogen with an efficiency of 80–90%. Lastly, this approach may lead the entire biomass pyrolysis process to be an overall carbon-neutral process.
- Authors:
-
- Georgia Inst. of Technology, Atlanta, GA (United States). School of Civil and Environmental Engineering
- Univ. of Tennessee, Knoxville, TN (United States). Bredesen Center for Interdisciplinary Research and Education
- Oak Ridge National Lab. (ORNL), Oak Ridge, TN (United States)
- Univ. of Tennessee, Knoxville, TN (United States). Bredesen Center for Interdisciplinary Research and Education; Oak Ridge National Lab. (ORNL), Oak Ridge, TN (United States)
- Georgia Inst. of Technology, Atlanta, GA (United States). School of Civil and Environmental Engineering; Oak Ridge National Lab. (ORNL), Oak Ridge, TN (United States)
- Publication Date:
- Research Org.:
- Oak Ridge National Lab. (ORNL), Oak Ridge, TN (United States)
- Sponsoring Org.:
- USDOE Office of Energy Efficiency and Renewable Energy (EERE), Sustainable Transportation Office. Bioenergy Technologies Office
- OSTI Identifier:
- 1430619
- Alternate Identifier(s):
- OSTI ID: 1548697
- Grant/Contract Number:
- AC05-00OR22725; DEAC05-00OR22725
- Resource Type:
- Accepted Manuscript
- Journal Name:
- Renewable Energy
- Additional Journal Information:
- Journal Volume: 125; Journal Issue: C; Journal ID: ISSN 0960-1481
- Publisher:
- Elsevier
- Country of Publication:
- United States
- Language:
- English
- Subject:
- 09 BIOMASS FUELS; Bio-oil; Pyrolysis oil; pH neutralization; Electrosorption; Capacitive deionization; Microbial electrolysis
Citation Formats
Park, Lydia Kyoung-Eun, Satinover, Scott J., Yiacoumi, Sotira, Mayes, Richard T., Borole, Abhijeet P., and Tsouris, Costas. Electrosorption of organic acids from aqueous bio-oil and conversion into hydrogen via microbial electrolysis cells. United States: N. p., 2018.
Web. doi:10.1016/j.renene.2018.02.076.
Park, Lydia Kyoung-Eun, Satinover, Scott J., Yiacoumi, Sotira, Mayes, Richard T., Borole, Abhijeet P., & Tsouris, Costas. Electrosorption of organic acids from aqueous bio-oil and conversion into hydrogen via microbial electrolysis cells. United States. https://doi.org/10.1016/j.renene.2018.02.076
Park, Lydia Kyoung-Eun, Satinover, Scott J., Yiacoumi, Sotira, Mayes, Richard T., Borole, Abhijeet P., and Tsouris, Costas. Sat .
"Electrosorption of organic acids from aqueous bio-oil and conversion into hydrogen via microbial electrolysis cells". United States. https://doi.org/10.1016/j.renene.2018.02.076. https://www.osti.gov/servlets/purl/1430619.
@article{osti_1430619,
title = {Electrosorption of organic acids from aqueous bio-oil and conversion into hydrogen via microbial electrolysis cells},
author = {Park, Lydia Kyoung-Eun and Satinover, Scott J. and Yiacoumi, Sotira and Mayes, Richard T. and Borole, Abhijeet P. and Tsouris, Costas},
abstractNote = {Neutralization of the bio-oil pH has been shown to generate a neutralized bio-oil aqueous phase (NBOAP) that includes most of the acidic components and a neutralized bio-oil organic phase (NBOOP) that includes hydrophobic organics, such as phenols. NBOOP can be used for fuel production, while NBOAP can be fed to microbial electrolysis cells (MECs) for hydrogen production. After pH neutralization, some organic acidic components remain in NBOOP. This work is focused on capturing acidic compounds from NBOOP through water extraction and electrosorption, and demonstrating hydrogen production via MECs. Capacitive deionization (CDI) is proven effective in capturing ions from NBOOP-contacted water and NBOAP via electrosorption. Captured acidic compounds enable the MEC application to effectively produce renewable hydrogen. Chemical oxygen demand (COD) removal of 49.2%, 61.5%, and 60.8% for 2, 4, and 10 g/L-anode/day loading were observed, corresponding to a total COD degradation of 0.19 g/L, 0.79 g/L, and 1.3 g/L, respectively. A maximum hydrogen productivity of 4.3 L-H2/L-anode/day was obtained. Major compounds in the water phase such as fatty acids, sugar derivatives, furanic and phenolic compounds were converted to hydrogen with an efficiency of 80–90%. Lastly, this approach may lead the entire biomass pyrolysis process to be an overall carbon-neutral process.},
doi = {10.1016/j.renene.2018.02.076},
journal = {Renewable Energy},
number = C,
volume = 125,
place = {United States},
year = {Sat Feb 17 00:00:00 EST 2018},
month = {Sat Feb 17 00:00:00 EST 2018}
}
Web of Science
Figures / Tables:
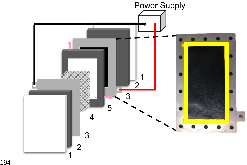 Figure 1: CDI cell with two half cells, each of which contains (1) a Plexiglas cover, (2) a viton gasket, (3) a titanium plate (current collector) with a carbon sheet, (4) a plastic mesh to keep the electrodes separated, and (5) a hollow middle viton gasket with syringe needles formore »
Figure 1: CDI cell with two half cells, each of which contains (1) a Plexiglas cover, (2) a viton gasket, (3) a titanium plate (current collector) with a carbon sheet, (4) a plastic mesh to keep the electrodes separated, and (5) a hollow middle viton gasket with syringe needles formore »
Figures / Tables found in this record:

 Search WorldCat to find libraries that may hold this journal
Search WorldCat to find libraries that may hold this journal