Insights into the Interconnection of the Electrodes and Electrolyte Species in Lithium–Sulfur Batteries Using Spatially Resolved Operando X-ray Absorption Spectroscopy and X-ray Fluorescence Mapping
- Technical Univ. of Munich, Garching (Germany). Chair of Technical Electrochemistry. Dept. of Chemistry. Catalysis Research Center
- SLAC National Accelerator Lab., Menlo Park, CA (United States). Stanford Synchrotron Radiation Lightsource
- Univ. of Amsterdam (Netherlands). Sustainable Materials Characterization. Van’t Hoff Inst. for Molecular Sciences
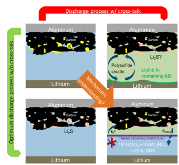
The lithium–sulfur (Li–S) battery chemistry has attracted great interest in the last decade because of its outstanding theoretical gravimetric energy density compared to the state-of-the-art lithium-ion battery technology. However, practically achieved energy density is still far below the theoretical value, even in small laboratory-scale batteries. The problems seen in laboratory-scale batteries will inevitably increase during scale-up to large application-format cells, as the electrolyte to active material (AM) ratio will need to be reduced in these cells to achieve high gravimetric energy density on cell-level basis. Our paper shows the unique possibility of X-ray fluorescence (XRF) mapping to visualize the spatial distribution of the AM inside operating Li–S batteries in all cell components [working electrode (WE), separator, and counter electrode (CE)]. Through a combination of operando XRF mapping and X-ray absorption spectroscopy, we show that unless self-discharge is efficiently prevented, the AM can completely dissolve and distribute throughout the cell stack within a time frame of 2 h, causing poor capacity retention. Finally, using a polysulfide diffusion barrier between the WE and the CE, we successfully suppress these processes and thereby establish a tool for examining the sealed cathode electrode compartment, enabling sophisticated studies for future optimization of the WE processes.
- Research Organization:
- SLAC National Accelerator Lab., Menlo Park, CA (United States); Technical Univ. of Munich, Garching (Germany); Univ. of Amsterdam (Netherlands)
- Sponsoring Organization:
- USDOE Office of Science (SC), Basic Energy Sciences (BES); Federal Ministry for Economic Affairs and Energy (BMWi) (Germany); Netherlands Organisation for Scientific Research (NWO)
- Grant/Contract Number:
- AC02-76SF00515; 03ET6045D; VIDI 723.014.010
- OSTI ID:
- 1471518
- Journal Information:
- Journal of Physical Chemistry. C, Vol. 122, Issue 10; ISSN 1932-7447
- Publisher:
- American Chemical SocietyCopyright Statement
- Country of Publication:
- United States
- Language:
- English
Web of Science
The Importance of Chemical Reactions in the Charging Process of Lithium-Sulfur Batteries
|
journal | January 2018 |
Deciphering the Reaction Mechanism of Lithium–Sulfur Batteries by In Situ/Operando Synchrotron‐Based Characterization Techniques
|
journal | March 2019 |
Similar Records
Direct observation of the redistribution of sulfur and polysufides in Li-S batteries during first cycle by in situ X-Ray fluorescence microscopy
Impact of Charge Voltage on Factors Influencing Capacity Fade in Layered NMC622: Multimodal X-ray and Electrochemical Characterization