De novo DNA synthesis using polymerase-nucleotide conjugates
- Joint BioEnergy Inst. (JBEI), Emeryville, CA (United States); Lawrence Berkeley National Lab. (LBNL), Berkeley, CA (United States); Technische Univ. Darmstadt, Darmstadt (Germany)
- Joint BioEnergy Inst. (JBEI), Emeryville, CA (United States); Lawrence Berkeley National Lab. (LBNL), Berkeley, CA (United States); Univ. of California, Berkeley, CA (United States)
- Joint BioEnergy Inst. (JBEI), Emeryville, CA (United States); Lawrence Berkeley National Lab. (LBNL), Berkeley, CA (United States)
- Joint BioEnergy Inst. (JBEI), Emeryville, CA (United States); Sandia National Lab. (SNL-CA), Livermore, CA (United States)
- Joint BioEnergy Inst. (JBEI), Emeryville, CA (United States); Lawrence Berkeley National Lab. (LBNL), Berkeley, CA (United States); USDOE Joint Genome Institute (JGI), Walnut Creek, CA (United States)
- Joint BioEnergy Inst. (JBEI), Emeryville, CA (United States); Lawrence Berkeley National Lab. (LBNL), Berkeley, CA (United States); Univ. of California, Berkeley, CA (United States); Technical Univ. of Denmark, Horsholm (Denmark)
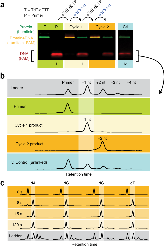
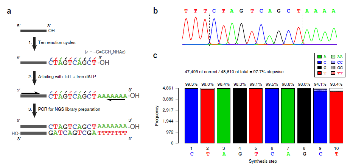
Oligonucleotides are almost exclusively synthesized using the nucleoside phosphoramidite method, even though it is limited to the direct synthesis of ~200 mers and produces hazardous waste. Here, we describe an oligonucleotide synthesis strategy that uses the template-independent polymerase terminal deoxynucleotidyl transferase (TdT). Each TdT molecule is conjugated to a single deoxyribonucleoside triphosphate (dNTP) molecule that it can incorporate into a primer. After incorporation of the tethered dNTP, the 3' end of the primer remains covalently bound to TdT and is inaccessible to other TdT-dNTP molecules. Cleaving the linkage between TdT and the incorporated nucleotide releases the primer and allows subsequent extension. We demonstrate that TdT-dNTP conjugates can quantitatively extend a primer by a single nucleotide in 10-20 s, and that the scheme can be iterated to write a defined sequence. Furthermore, this approach may form the basis of an enzymatic oligonucleotide synthesizer.
- Research Organization:
- Lawrence Berkeley National Laboratory (LBNL), Berkeley, CA (United States)
- Sponsoring Organization:
- USDOE Office of Science (SC), Biological and Environmental Research (BER)
- Grant/Contract Number:
- AC02-05CH11231
- OSTI ID:
- 1461176
- Journal Information:
- Nature Biotechnology, Vol. 36, Issue 7; ISSN 1087-0156
- Publisher:
- Springer NatureCopyright Statement
- Country of Publication:
- United States
- Language:
- English
Web of Science
Similar Records
Automatable process for sequencing nucleotide
Incorporation of dA opposite N3-ethylthymidine terminates in vitro DNA synthesis