Hydrogenation properties of lithium and sodium hydride – closo -borate, [B 10 H 10 ] 2− and [B 12 H 12 ] 2− , composites
- Center for Materials Crystallography, Interdisciplinary Nanoscience Center (iNANO) and Department of Chemistry, Aarhus University, 8000 Aarhus C, Denmark
- Chemistry, Combustion, and Materials Center, Sandia National Laboratories, Livermore, USA
- Department of Chemistry and Interdisciplinary Nanoscience Center (iNANO), Aarhus University, 8000 Aarhus C, Denmark
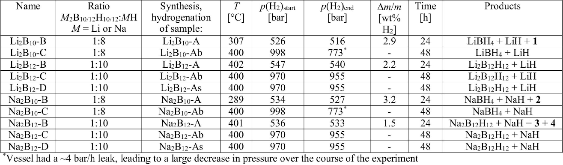
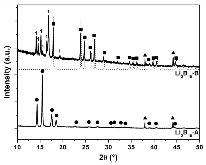
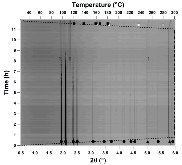
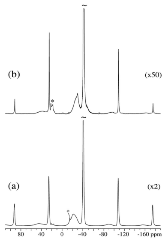
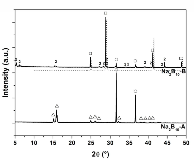
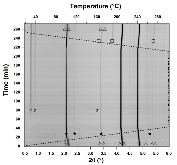
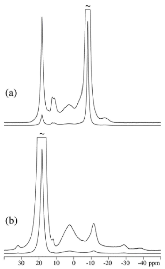
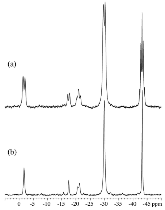
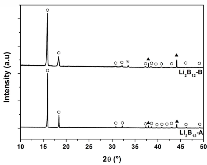
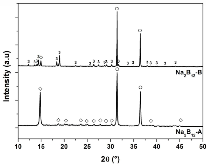
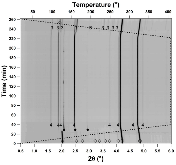
The hydrogen absorption properties of metal closo-borate metal hydride composites, M2B10H10– 8MH and M2B12H12–10MH, M = Li or Na, are studied under high hydrogen pressures to understand the formation mechanism of metal borohydrides. The hydrogen storage properties of the composites have been investigated by in-situ synchrotron radiation powder X-ray diffraction at p(H2) = 400 bar and by ex-situ hydrogen absorption measurements at p(H2) = 526 to 998 bar. The in-situ experiments reveal the formation of crystalline intermediates before metal borohydrides (MBH4) are formed. The M2B12H12–10MH (M = Li and Na) systems show no formation of the metal borohydride at T = 400 °C and p(H2) = 537 to 970 bar. 11B MAS NMR of the M2B10H10– 8MH composites reveal that the molar ratio of LiBH4 or NaBH4 and the remaining B species is 1:0.63 and 1:0.21, respectively. Solution and solid-state 11B NMR spectra reveal new intermediates with a B:H ratio close to 1:1. Our results indicate that the M2B10H10 (M = Li, Na) salts display a higher reactivity towards hydrogen in the presence of metal hydrides compared to the corresponding [B12H12]2- compounds, which represents an important step towards understanding the factors that determine the stability and reversibility of high hydrogen capacity metal borohydrides for hydrogen storage.
- Research Organization:
- Sandia National Lab. (SNL-NM), Albuquerque, NM (United States); Sandia National Lab. (SNL-CA), Livermore, CA (United States)
- Sponsoring Organization:
- USDOE National Nuclear Security Administration (NNSA); USDOE Office of Energy Efficiency and Renewable Energy (EERE), Sustainable Transportation Office. Hydrogen Fuel Cell Technologies Office
- Grant/Contract Number:
- AC04-94AL85000; NA0003525
- OSTI ID:
- 1470579
- Alternate ID(s):
- OSTI ID: 1429663; OSTI ID: 1440264; OSTI ID: 1444088
- Report Number(s):
- SAND-2017-11502J; SAND-2018-6006J; PPCPFQ
- Journal Information:
- Physical Chemistry Chemical Physics. PCCP (Print), Journal Name: Physical Chemistry Chemical Physics. PCCP (Print) Vol. 20 Journal Issue: 23; ISSN 1463-9076
- Publisher:
- Royal Society of Chemistry (RSC)Copyright Statement
- Country of Publication:
- United Kingdom
- Language:
- English
Web of Science
Hydrogenation properties of lithium and sodium hydride – closo -borate, $\mathrm{[B_{10}H_{10}]^{2−}$ and $[B_{12}H_{12}]^{2−}}$ , composites
|
text | January 2018 |
Quantitative Assessment of B−B−B, B−H b −B, and B−H t Bonds: From BH 3 to B 12 H 12 2−
|
journal | July 2019 |
A fleeting glimpse of the dual roles of SiB 4 in promoting the hydrogen storage performance of LiBH 4
|
journal | January 2019 |
Potassium octahydridotriborate: diverse polymorphism in a potential hydrogen storage material and potassium ion conductor
|
journal | January 2019 |
Similar Records
Complex high-temperature phase transitions in Li{sub 2}B{sub 12}H{sub 12} and Na{sub 2}B{sub 12}H{sub 12}
Understanding and Mitigating the Effects of Stable Dodecahydro- closo -dodecaborate Intermediates on Hydrogen-Storage Reactions